MBB 222 Lecture Notes - Lecture 3: Aromaticity, Atomic Orbital, Aliphatic Compound
11 views2 pages
20 Dec 2018
School
Department
Course
Professor
Document Summary
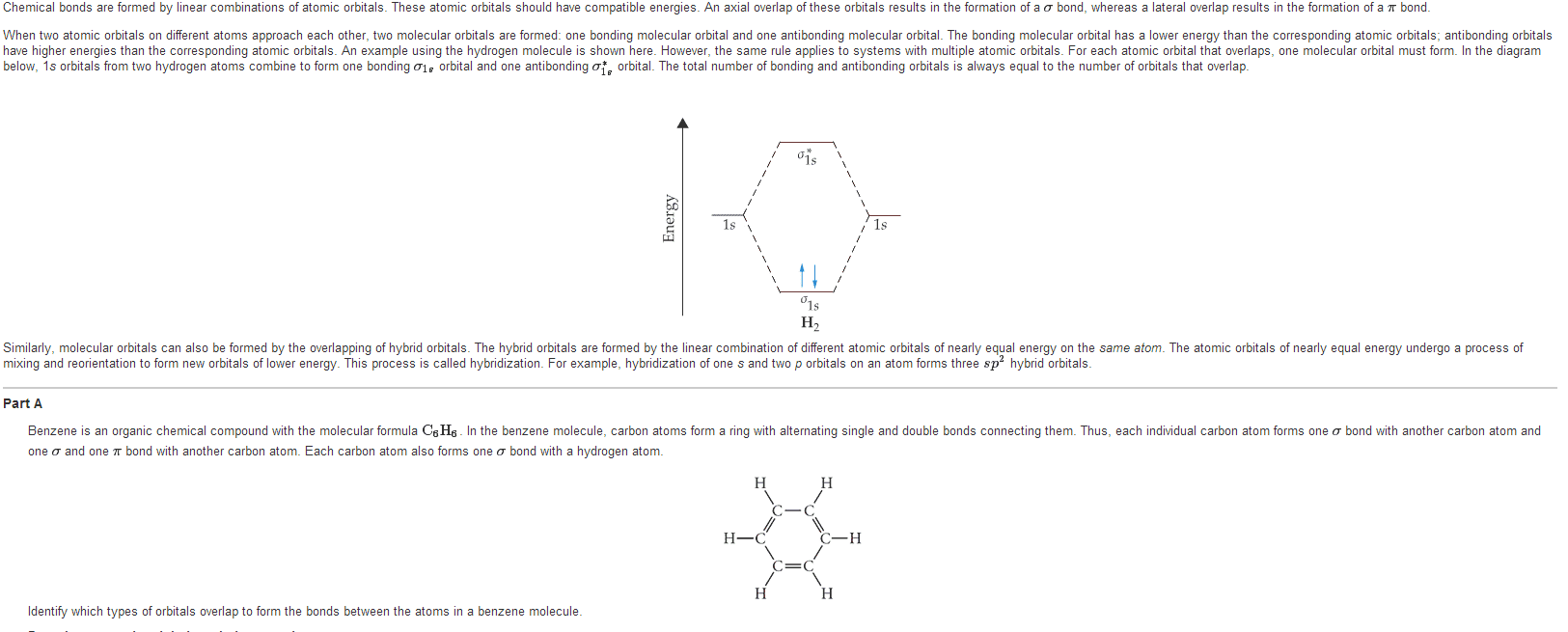
An important class of compounds in chemistry and biology is the aromatic compounds. These cyclic ring-shaped molecules have alternating single and double bonds, resulting in delocalization of their electrons to form a (pi) electron cloud. Compare benzene, an aromatic molecule, with cyclohexane, an aliphatic molecule. Cyclohexane: cyclized 6-carbon ring with carbons linked by single bonds. Benzene: cyclized 6-carbon ring with carbons linked by alternating single and double bonds. Each c has one h rigid, planar structure. Aromatic compound - electrons are delocalized in a cloud - resonance . Has unique properties due to resonance very stable (not reactive) Each electron is shared by all six carbons - the electrons are delocalized. While many compounds have bonds and exhibit resonance, only cyclic molecules that form a closed loop of overlapping orbitals are aromatic. Aromatic compounds usually have distinctive physical properties that affect their interactions with each other and with other molecules.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers