BIOCH200 Lecture Notes - Schiff Base, Lone Pair, Histidine
Document Summary
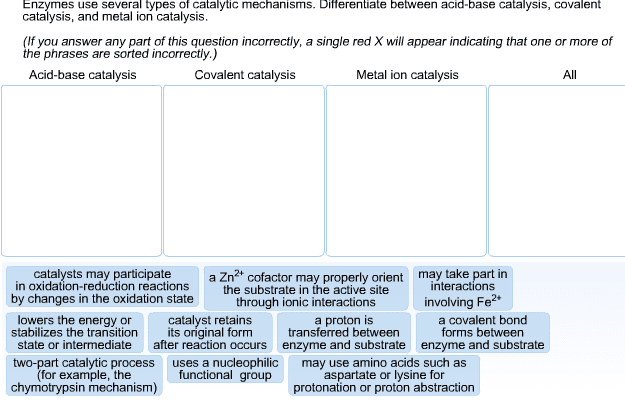
Bioch 200 enzymes (con"t) (march 7, 2014) some overlap. *chemical reactions only occur if the reactants are in the right orientation in regards to each other: proximity and orientation effects of catalysis, substrate binding to enzyme is in a certain orientation. Bringing together substrates in this way greatly increases the rate of reaction: *when enzymes catalyze reactions, they may donate/accept protons but this is temporary the enzyme will always return back to its original state. *amino acids that take part in acid-base catalysis. Act as an acid or a base depending on their state of protonation: aspartate (or aspartic acid, glutamate (or glutamic acid, histidine, lysine, cysteine, tyrosine. *amino acids that take part in nucleophillic catalysis (aka covalent catalysis). These amino acids can act as nucleophiles when they get deprotonated because they then have a lone pair of electrons, something that is needed for nucleophillic attack: serine/tyrosine, cysteine, lysine, histidine, schiff base = imine .