CHEM101 Lecture Notes - Lecture 9: Electron Configuration, Valence Electron, Transition Metal
CHEM101 verified notes
9/41View all
8

CHEM101 Lecture 8: January 23: effective charge, screening, orbitals, relative energy of orbitals, and electron configuration of the elements
9

CHEM101 Lecture Notes - Lecture 9: Electron Configuration, Valence Electron, Transition Metal
10

CHEM101 Lecture Notes - Lecture 10: Van Der Waals Radius, Ionic Radius, Periodic Trends
Document Summary
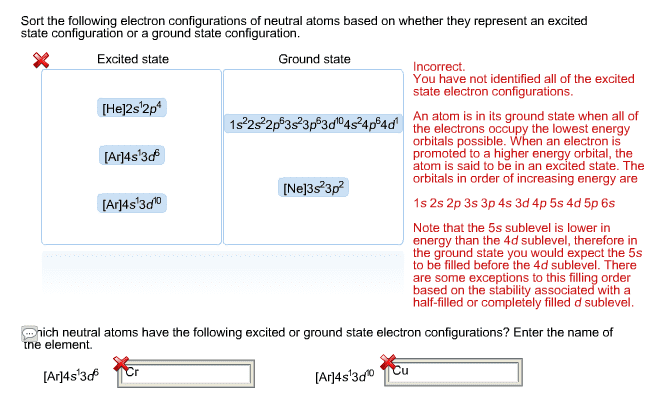
January 25th: electron configurations of the elemets, of ions, of transition metals, predicting atomic electronic configurations of metals, preiodic trends in atomic properties, valence electrons and zeff. In most cases the general rules are followed, which is that the energy in 4s is larger than 3d and 5s has a higher energy level than 4d. But, transition metals have some exceptions such as chromium which has an electron configuration of. Cr:[ar]4s^13d^5 where you can see that the s orbital isn"t full because a full s orbital would have 2 electrons. Another exception is calcium with an electron configuration of. These exceptions have lower energy in the s orbital because they are not completely full. Main group metal ions tend to lose electrons so they can obtain a noble gas configuration. An example of this would be na na+ + e- . Main group nonmetal ions are quite the opposite, they gain an electron to obtain a noble gas configuration.