CHMB42H3 Lecture Notes - Nitronium Ion, Methylene Group, Electrophilic Aromatic Substitution
Document Summary
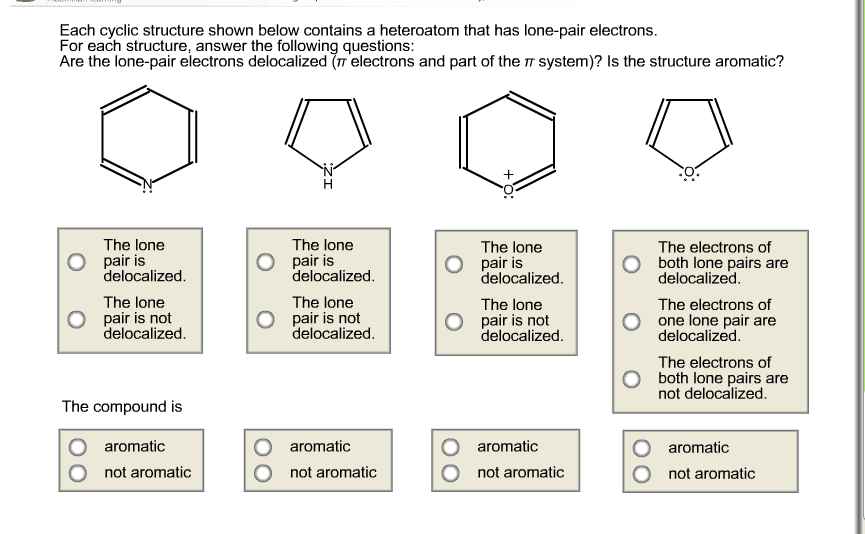
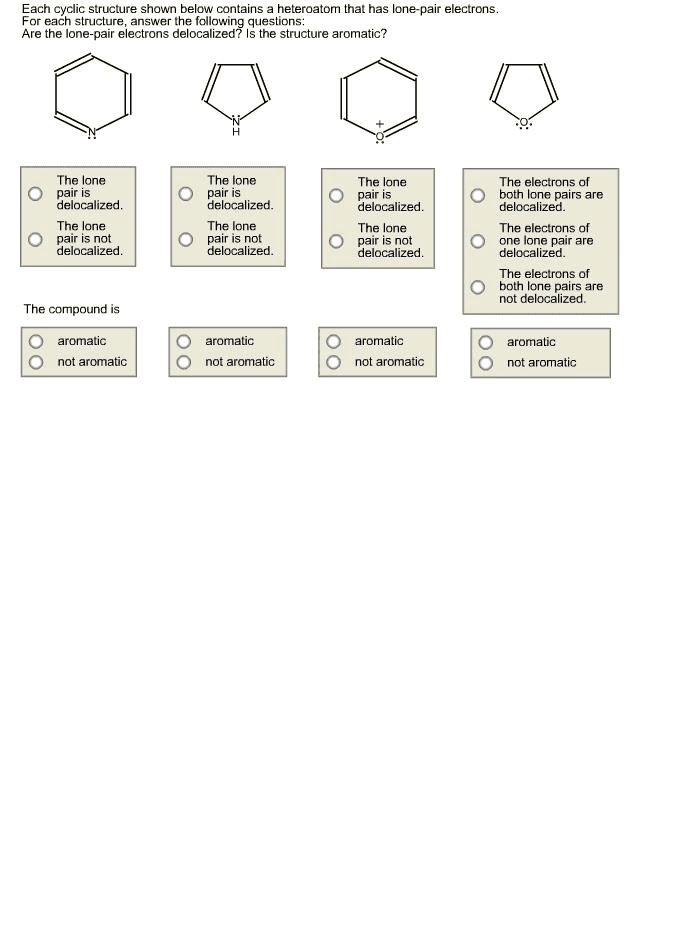
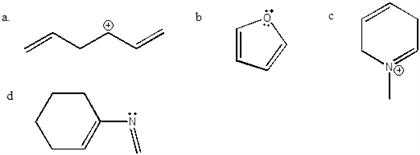
Benzene is very stable: has very high delocalization energy. Recall: delocalization energy is a measure of how much more a compound with delocalized electrons is than it would be with localized electrons. More so than other compounds with delocalized energies. Aromatic compounds compounds with unusually large resonance energies: these are particularly stable. The two criteria for aromaticity: 1. Must have an uninterrupted cyclic cloud of electrons (called a cloud) above and below the plane of the molecule. Every atom in the ring must have a p orbital. Each p orbital must overlap with the p orbitals on either side of it: thus, the molecule must be planar, 2. The cloud must contain an odd number of pairs of electrons. For a compound to be aromatic, it must be cyclic and planar and have an uninterrupted cloud of electrons. The cloud must contain an off number of pairs of electrons. An odd number of pairs of electrons.