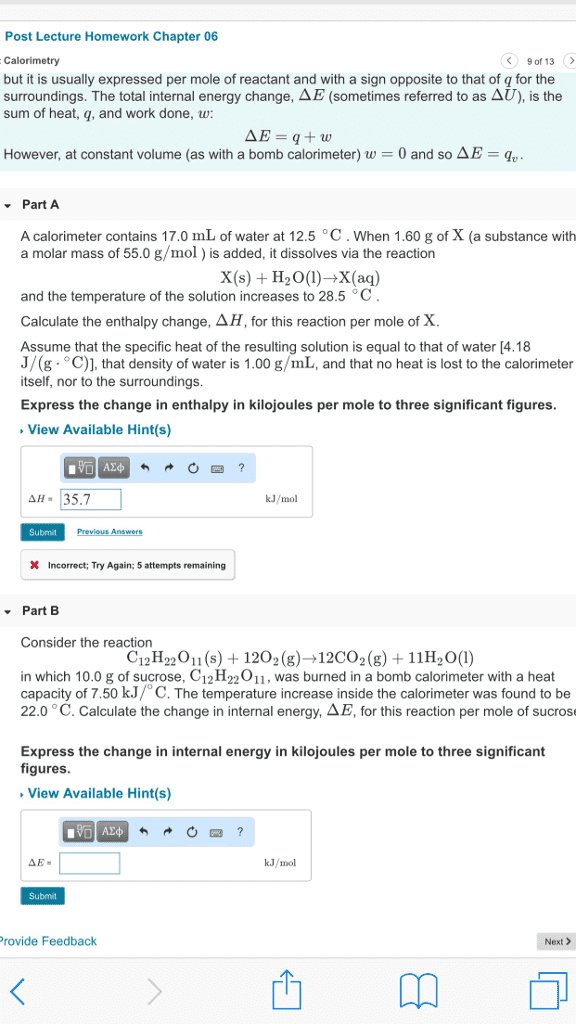
Calorimetry is a method used to measure changes inenthalpy, or heat, that occur during chemical processes. Two commoncalorimeters are constant-pressure calorimeters and constant-volume(or "bomb") calorimeters. Bomb calorimeters are used to measurecombustion and other gas-producing reactions, in which the reactionis observed in a strong, sealed vessel. A simple constant-pressurecalorimeter can be made from a foam coffee cup and a thermometer,in which energy changes in a reaction are observed via the changein temperature of the solution in the cup. The idea behindcalorimeters is that if they are sufficiently insulated from theoutside environment, any energy gained or lost in the chemicalreaction will be directly observable as a temperature and/orpressure change in the calorimeter.

Calorimetry is a method used to measure changes in enthalpy, or heat, that occur during chemical processes. Two common calorimeters are constant-pressure calorimeters and constant-volume (or "bomb") calorimeters. Bomb calorimeters are used to measure combustion and other gas-producing reactions, in which the reaction is observed in a strong, sealed vessel. A simple constant-pressure calorimeter can be made from a foam coffee cup and a thermometer, in which energy changes in a reaction are observed via the change in temperature of the solution in the cup. The idea behind calorimeters is that if they are sufficiently insulated from the outside environment, any energy gained or lost in the chemical reaction will be directly observable as a temperature and/or pressure change in the calorimeter.
Show transcribed image text Calorimetry is a method used to measure changes in enthalpy, or heat, that occur during chemical processes. Two common calorimeters are constant-pressure calorimeters and constant-volume (or "bomb") calorimeters. Bomb calorimeters are used to measure combustion and other gas-producing reactions, in which the reaction is observed in a strong, sealed vessel. A simple constant-pressure calorimeter can be made from a foam coffee cup and a thermometer, in which energy changes in a reaction are observed via the change in temperature of the solution in the cup. The idea behind calorimeters is that if they are sufficiently insulated from the outside environment, any energy gained or lost in the chemical reaction will be directly observable as a temperature and/or pressure change in the calorimeter.