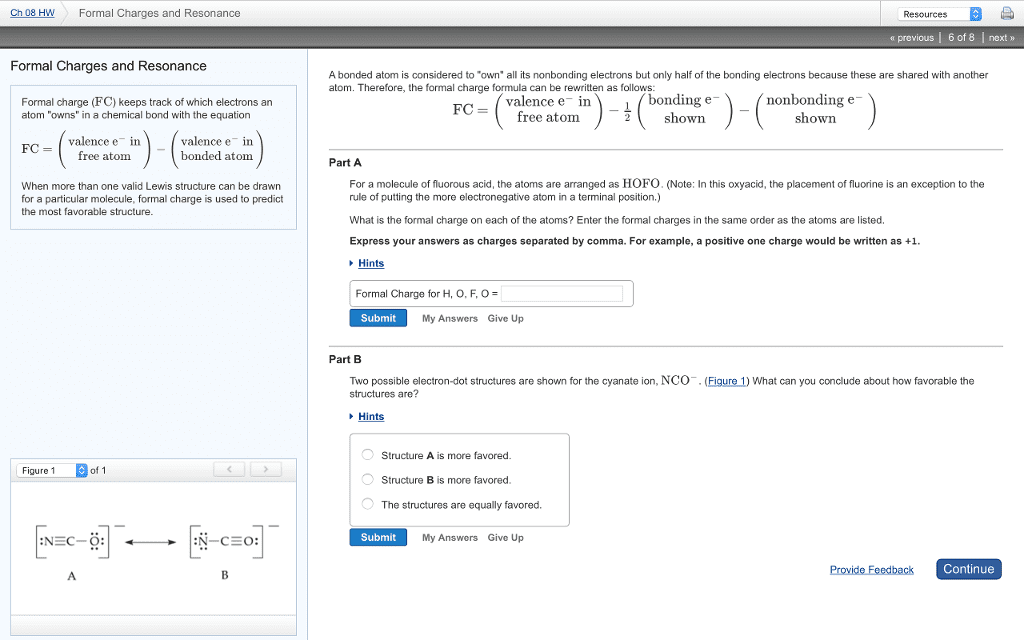
Ch 08 HW Formal Charges and Resonance Resources a previous | 6 of 8 Inext » Formal Charges and Resonance A bonded atom is considered to "own" all its nonbonding electrons but only half of the bonding electrons because these are shared with another atom. Therefore, the formal charge formula can be rewritten as follows valence e in bonding e nonbonding e- Formal charge (FC) keeps track of which electrons an atom "owns" in a chemical bond with the equation FCfree atom shown shown valence e in free atom valence e in bonded atom FC Part A For a molecule of fluorous acid, the atoms are arranged as HOFO. (Note: In this oxyacid, the placement of fluorine is an exception to the When more than one valid Lewis structure can be drawn for a particular molecule, formal charge is used to predict the most favorable structure. rule of putting the more electronegative atom in a terminal position.) What is the formal charge on each of the atoms? Enter the formal charges in the same order as the atoms are listed. Express your answers as charges separated by comma. For example, a positive one charge would be written as +1 Hints Formal Charge for H, O, F, O = Submit My Answers Give Up Part B Two possible electron-dot structures are shown for the cyanate ion, NCO. (Figure 1) What can you conclude about how favorable the structures are? Hints Structure A is more favored Structure B is more favored. The structures are equally favored. Figure 1 of 1 NEC-OEN-CEO: Submit My Answers Give Up Continue





