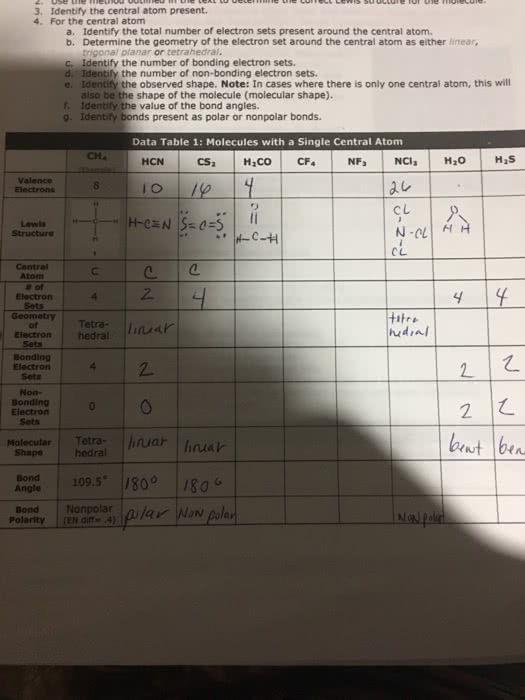
molecules with a single central ato
3. Identify the central atom present. 4. For the central atom a. Identify the total number of electron sets present around the central atom. b. Determine the geometry of the electron set around the central atom as either linear, trigonal planar or tetrahedral. c Identify the number of bonding electron sets. d. Identify the number of non-bonding electron sets. e. Identify the observed shape. Note: In cases where there is only one central atom, this will also be the shape of the molecule (molecular shape). f. Identify the value of the bond angles g. Identify bonds present as polar or nonpolar bonds. Data Table 1: Molecules with a Single Central Atom dL.O ãã CH4 HCN CS2 H2CO CF Valence CU ì N-01 | C L Central Atom #of 4 Sets Tetra Electron hedral of Bonding Electron 2. Bonding 2 Molecular Tetra uar nakr hedra Angle Bond Nonpolar |u/ar Now paler Polarity (EN dIt4