CHM135H1 Lecture Notes - Reaction Rate Constant, Rate Equation
48 views1 pages
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related Documents
Related Questions
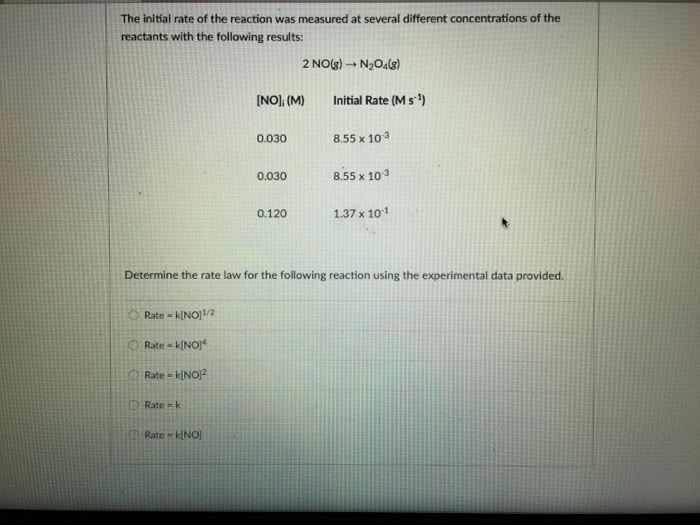
The initial rate of the reaction was measured at several different concentrations of the reactants with the following results:
2 NO(g) â N2O4(g)
| [NO]i (M) | Initial Rate (M s-1) |
| 0.030 | 8.55 x 10-3 |
| 0.030 | 8.55 x 10-3 |
| 0.120 | 1.37 x 10-1 |
Determine the rate law for the following reaction using the experimental data provided.
A) Rate = k[NO]1/2
B) Rate = k[NO]2
C)Rate = k[NO]4
D) Rate = k
E)Rate = k[NO]
I have no idea how to even approach this :(
this was all the information that was given