CHM135H1 Lecture Notes - Iodine Monochloride, Sodium Bicarbonate, Sodium Carbonate

90
CHM135H1 Full Course Notes
Verified Note
90 documents
Document Summary
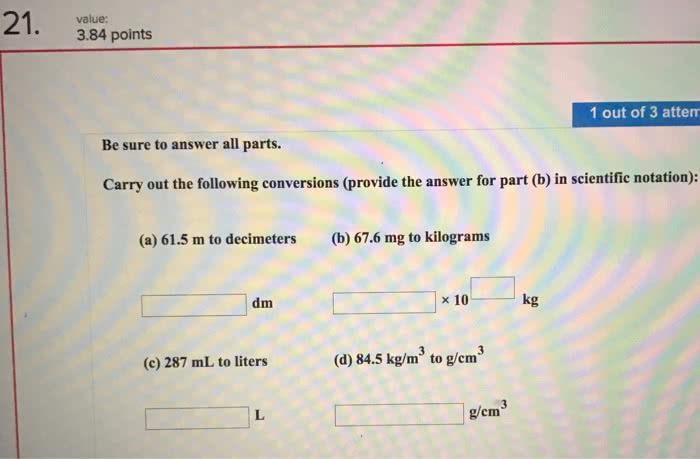
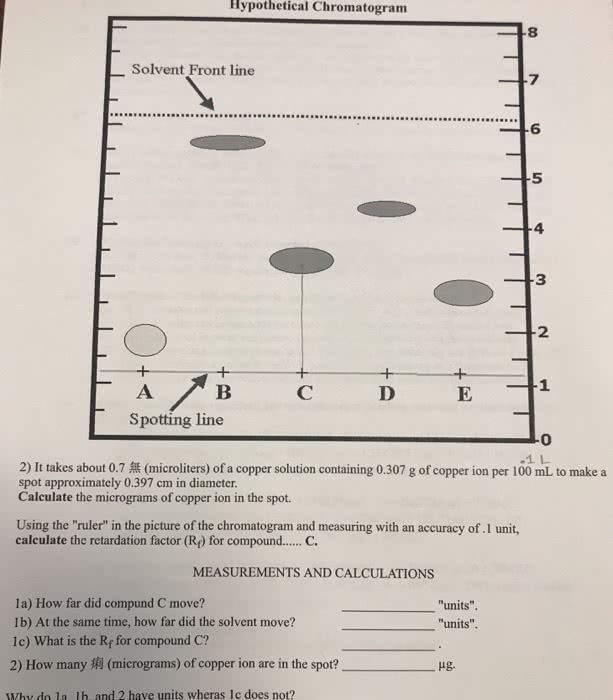
Units matter before si units, units were body-derived (ie. feet) Si units allow for worldwide standardization, so scientists can communicate without misunderstandings. One-tenth of a meter is a decimetre. One-tenth of a decimetre is a centimetre. Must be comfortable with scientific notation, and able to use it on a calculator. Must know how to convert between the multiples (x unit b / y unit a) is the conversation factor the equation gets you the measurement in unit b the two unit a s cancel out. Note that in the above equation, all units cancel out except for micrograms. Formal solution to ex: any mass above 86. 4 micrograms violates the standard. Measurement ranges between 2. 72 to 2. 74 cm. A has 4 significant digits, because the two zeros count. B the 0. 0 do not count as two significant digits, therefore 5 significant digits. C has 2 significant digits, see a for explanation.