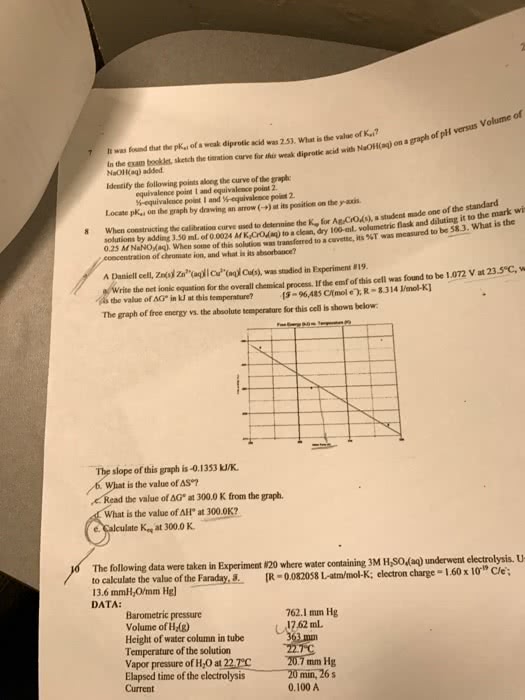
It was found that the pk_a1 of a weak diprotie acid was 2.53, What is the value of K_a1? In the exam booklet, sketch the titration curve for this weak diprotic acid with NaOH(aq) on a graph of pH versus Volume of NaOH(aq) added. Identify the following points along the curve of the graph: equivalence point 1 and equivalence point 2 equivalence point 1 and 1/2-equivalence point 2. Locate pK_a1 on the graph by drawing an arrow (rightarrow)at its position on the y-axis. When constructing the calibration curve used to determine the K_ for Ag_ CrO_ (s), a student made one of the standard solutions by adding 3.50 mL of 0.0024 MK_2CrO_ (aq) to a clean, dry 100-mLvolumetric flask and diluting it to the mark wi 0.25 M NaNO_ (aq). When some of this solution was transferred to a cuvette, its %T was measured to be 58.3. What is the concentration of chromate ion, and what is its absorbance? A Daniell cell, Z (s)|Zn^ (aq)||Ca^2+ (aq)|Cu(s), was studied in Experiment #19. write the net ionic equation for the overall chemical process. If the emf of this cell was found to be 1.072 V at 23 5 degree C, w is the value of G in kJ at dhis temperature? -96,485 Ctmol e), delta G degree in kJ at this temperature? [ =96,485 C/(mol ): R = 8.314 J/mol K] The graph of free energy vs. the absolute temperature for this cell is shown below: The slope of this graph is-0.1353 kJ/K. b. What is the value of delta S degree? c. Read the value of delta G degree at 300.0 K from the graph. d. What is the value of delta H degree at 300.0K? e. Calculate K_ q at 300.0 K. The following data were taken in Experiment #20 where water containing 3M H_2 SO_4 (aq) underwent electrolysis. U to calculate the value of the Faraday, [R = 0.082058 L-atm/mol-K: electron charge = 1.60 times 10^-19 C/e: 13.6 mmH_2O/mm Hg]