Chemistry 1027A/B Lecture Notes - Lecture 5: Rate Equation, Chemical Kinetics, Reaction Rate
Document Summary
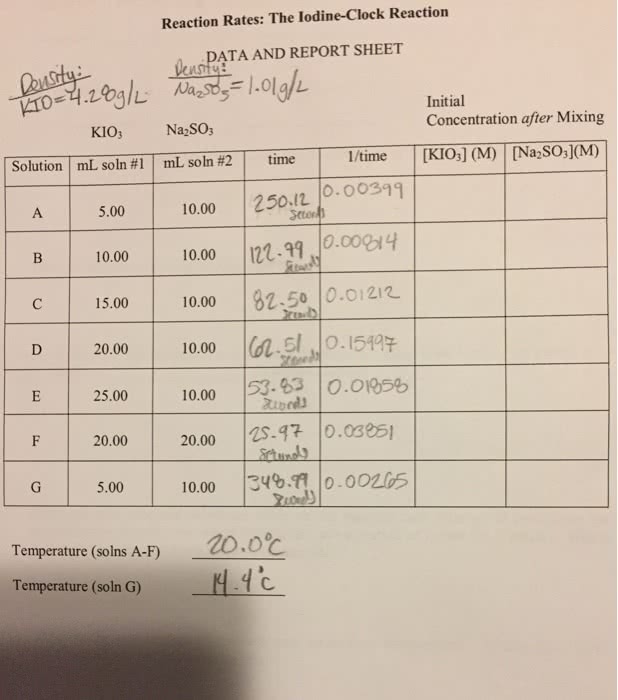
Chemical kinetics: study of how quickly a reaction will proceed (reaction rate) and factors affecting that rate. Speed at which a reaction takes place governed by several factors: what the reactants are, and their concentrations, temperature. Reactions tend to proceed faster at higher temperatures: catalyst presence. Rate of chemical reaction: positive quantity usually defined by comparing change in product or reactant over time, rate of product made, rate of reactant consumed. Summary aa + bb cc + dd. Effect of concentration on reaction described using rate laws aa + bb products. Rate of reaction (rate law) is given by: Rate = k[a]x[b]y k specific rate constant for reaction at given temperature. [a] & [b] are concentration of components a & b in m (moll-1) x & y are the orders for each reactant and can only be found experimentally. Exponents values are usually whole numbers determine order of reactant: don"t correlate with coefficients of reaction, x = 2.