BIOL 2020 Lecture Notes - Lecture 6: Globin, Proline, Transport Protein
Lecture 6 – Hemoglobin and Protein Structure
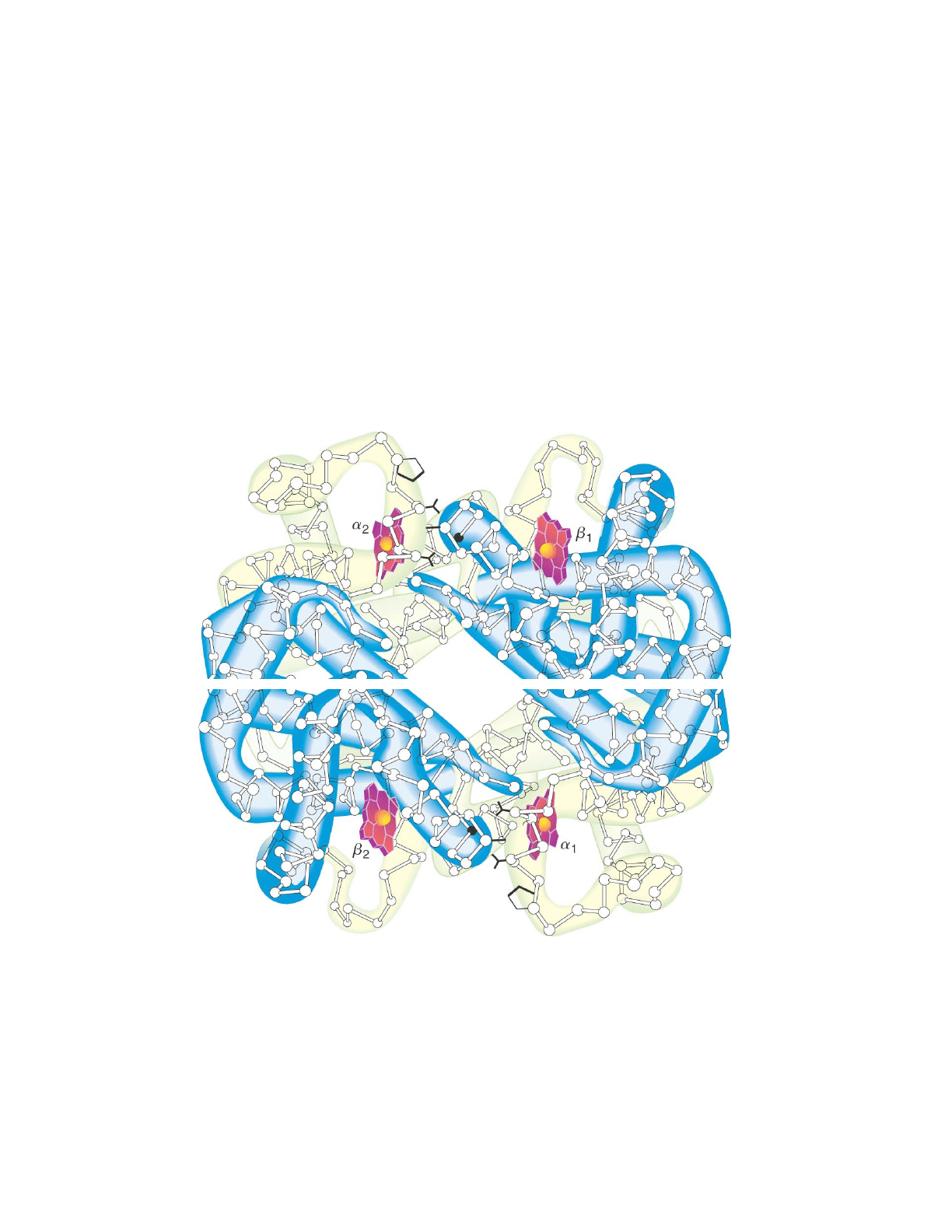
Hemoglobin
Abundant present in your blood
Exhibits quaternary structures
Transport protein
Oxygen carrying molecule of vertebrates; transports it to
peripheral targets around the body
Heterotypic
o2 a-subunits: a1 and a2
o2 B-subunits: B1 and B2
odon’t necessarily correspond to secondary structure of
hemoglobin
the subunits are referred to as globin fold
blue dimer (closer to us) is a1B2 dimer
the two dimers come together to form a tetromer
secondary structure of a globin fold consists of a-helices little
barrels
for each globin fold 8 a-helices at the end there’s a proline
that folds back and continue
Document Summary
Oxygen carrying molecule of vertebrates; transports it to peripheral targets around the body. Heterotypic: 2 a-subunits: a1 and a2, 2 b-subunits: b1 and b2, don"t necessarily correspond to secondary structure of hemoglobin the subunits are referred to as globin fold. Nomenclature for describing the a-helices beginning from n- terminus (a-alpha helix), goes up to h (8th fold) individually within a-helices, you can refer to speciic residue. 3rd aa within the 1st a-helix is the a-3 residue. F-8 (f helix and 8th residue) of each globin subunits. Helps stabilize the heme within the globin subunit of hemoglobin: distal hisidine makes a covalent bond with the iron. In absence of oxygen, it maintains the +2 state of oxygen and prevents it from becoming a ferric iron (+3) Hemoglobin binds to the oxygen (ligand) in 4 sites corresponding to the 4 hemes and 4 subunits looking at how well hemoglobin and myoglobin bind to oxygen o.

