CHEM 1000 Lecture Notes - Lecture 18: Pauli Exclusion Principle
Document Summary
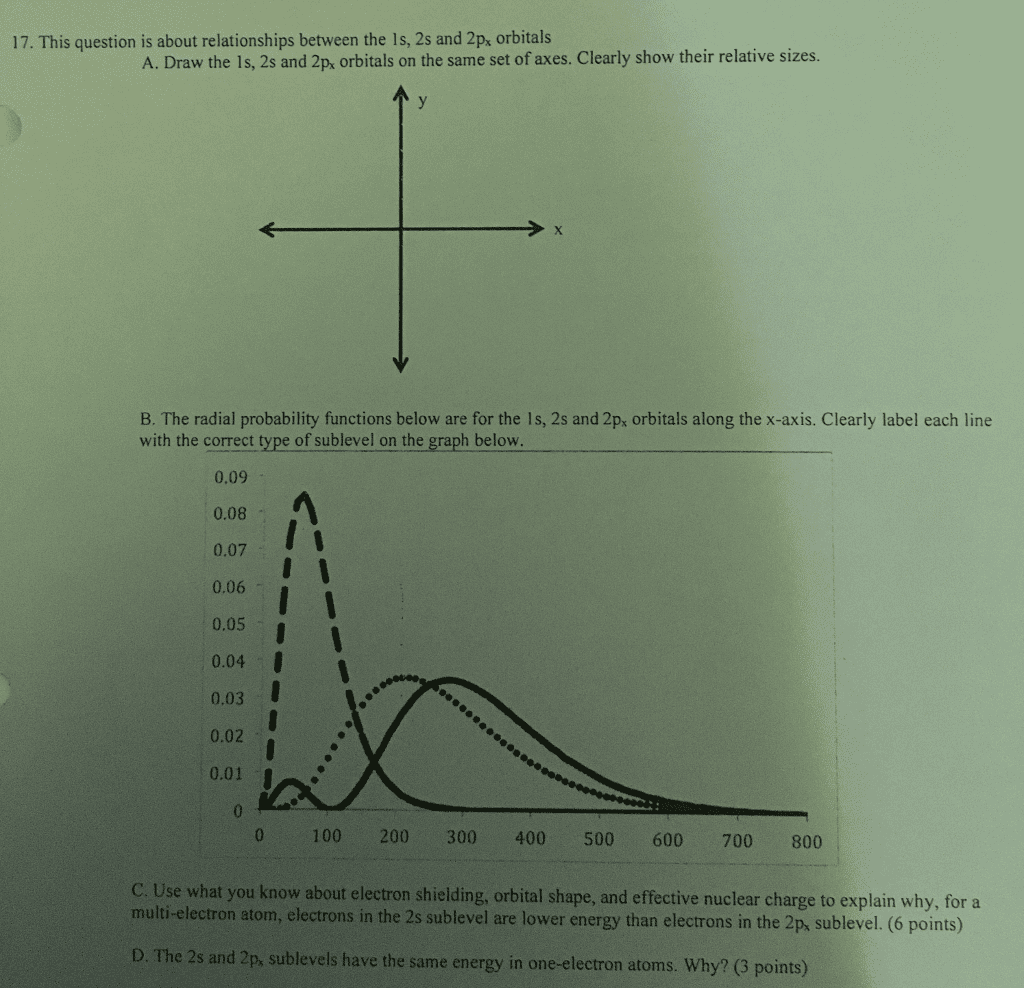
S(cid:272)h(cid:396) dinge(cid:396)"s e(cid:395)uation for a multi-electron atom has additional terms for repulsion between electrons. We can consider one electron at a time, in an environment established by nucleus & other electrons. Solving the schr dinger equation for this system gives hydrogen-like orbitals: the same shape, different sizes. At large distances from nucleus this works fine, however at close distances we see effects b/c shielding is not done by only spherical orbitals. Return to radial probability distribution for an electron in an orbital. (the probability multiplied by the surface area at distance r) Example: assume there are 2 electrons in 1s orbital & one electron in either the 2s or 2p orbital. Peak in e- density at around 0 means electron in 2s orbital can penetrate closer to nucleus than 2p. Nuclear attraction at closer distance means 2s e- is held tighter than 2p e- , so the 2s orbital will be at lower energy.