CHEM 2152 Lecture Notes - Lecture 4: Diethyl Ether, Sodium Sulfate, Sodium Bicarbonate

c. aasorption
2. Why is anhydrous sodium sulfate added to the organic solution remaining
after the extractions with 6 M HCl and 6 M NaOH?
3. Explain why swirling of a solution and its drying agent hastens the drying
process.
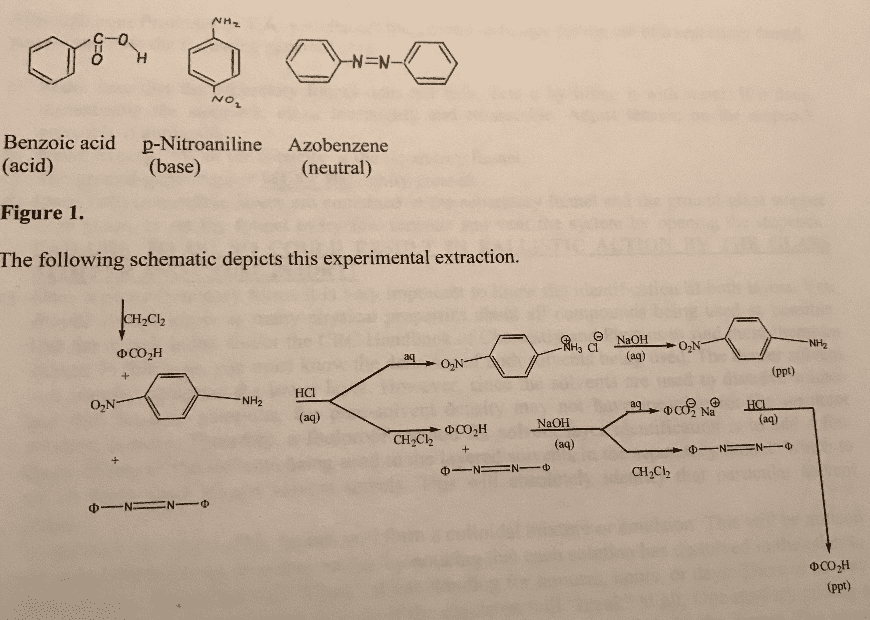
4. On the basis of what you learned in this experiment, plus the information
that phenol is soluble in aqueous sodium hydroxide but not in aqueous
sodium bicarbonate, Na HCO3, solution, whereas benzoic acid is soluble in
both solutions, show how you could separate a mixture containing phenol,
benzoic acid, p-nitroaniline, and anthracene.
5. a. Given 400 mL of an aqueous solution containing 12 g of compound A,
how many grams of A could be removed from the solution by a single
extraction with 200 mL of diethyl ether?
b. How many total grams of A can be removed with three successive
extractions of 67 mL each? (Assume the distribution coefficient for A in
diethyl ether/water is equal to 3.0.)
Note; In solving problems of this type, one should recognize that Equation
(5.3) applies to the situation pertaining after equilibrium has been reached.
For example, a practical form of the equation is shown below, where a =
grams of A originally present in water (S’) and x = grams of A present in
diethyl ether (5) after extraction.
X ^ m Lof5'
a - x mLofS
6. 8. The oKa of benzoin nriH is d IQ .Qhoiu
Document Summary
Note; in solving problems of this type, one should recognize that equation (5. 3) applies to the situation pertaining after equilibrium has been reached. For example, a practical form of the equation is shown below, where a = grams of a originally present in water (s") and x = grams of a present in diethyl ether (5) after extraction. X ^ m l o f5 " a - x m l ofs: 8. The oka of benzoin nrih is d iq . qhoiu. 1 4 8 ch a t t i r s extraction. Note: in solving problems of this type, one should recognize that equation (5. 3) applies to the situation pertaining after equilibrium has been reached. For example, a practical form of the equation is shown below, where a = grams of a originally present in water (s") and x = grams of a present in diethyl ether (s) after extraction.