CHEM 1A Lecture Notes - Lecture 6: Emission Spectrum, Empirical Formula, Magnetic Quantum Number
45 views2 pages

44
CHEM 1A Full Course Notes
Verified Note
44 documents
Document Summary
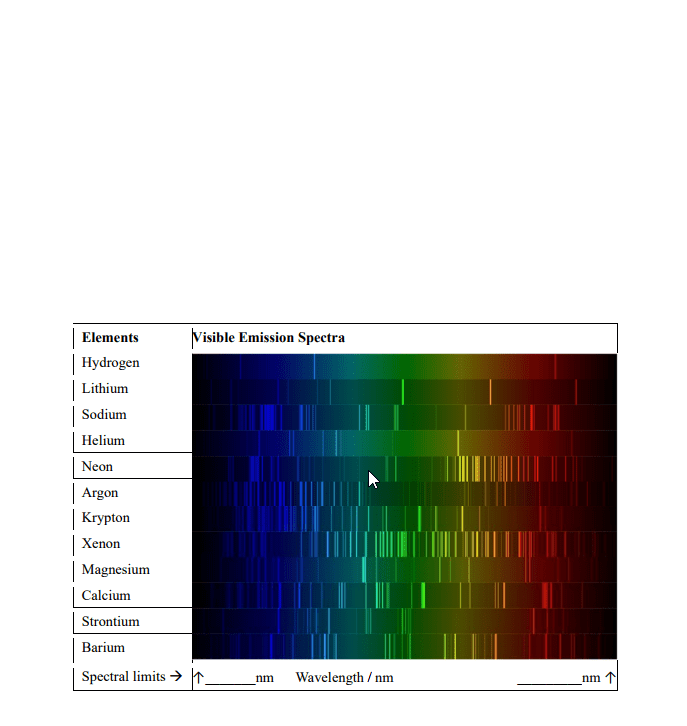
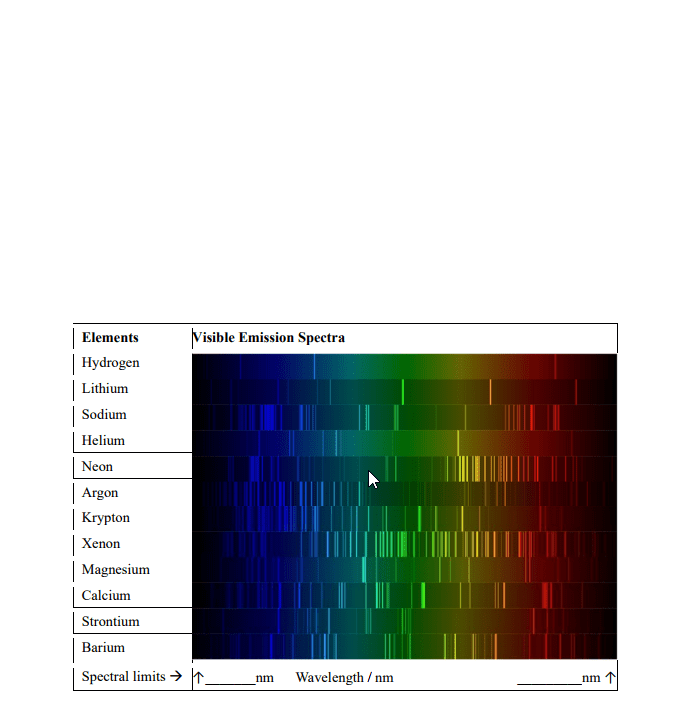
Contains no gaps and all wavelengths in the range are observed absorption spectrum: consists of dark lines on bright background. Observes what wavelengths are missing due to absorption by the sample. Electron move from one lower level to a higher level. Atom absorb energy = emit this energy as light. Light pass through a prism creates emission spectrum. If sample emitting light contains only one atom, known as atomic emission spectrum. Each atom has its own emission spectrum. De broglie"s relation: faster electron moves, smaller the wavelength is. = h / mv = h / p. Empirical formula describing the pattern of the visible wavelengths of the emission spectrum. The uncertainty principle: can never observe simultaneously the interference pattern and determine which slit the electron goes through. Can"t measure both velocity and position at same time. Principle quantum number (n): integer that determines overall size and energy of an orbital.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232