CHEM 1127Q Lecture Notes - Lecture 4: Isotopes Of Iron, Natural Abundance, Unified Atomic Mass Unit
CHEM 1127Q verified notes
4/4View all
Document Summary
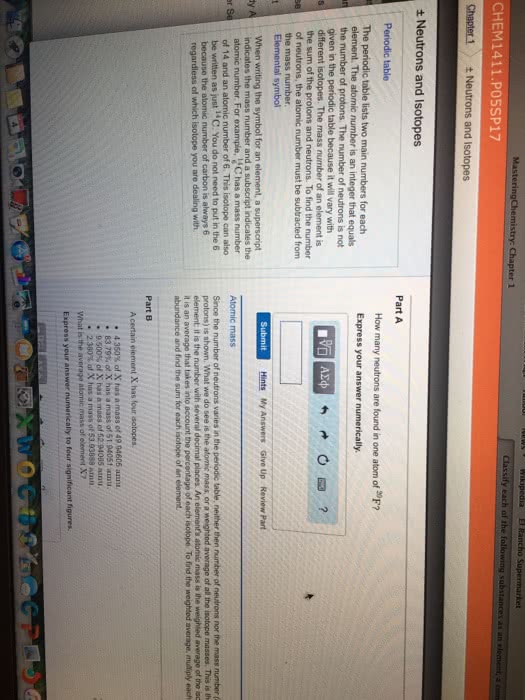
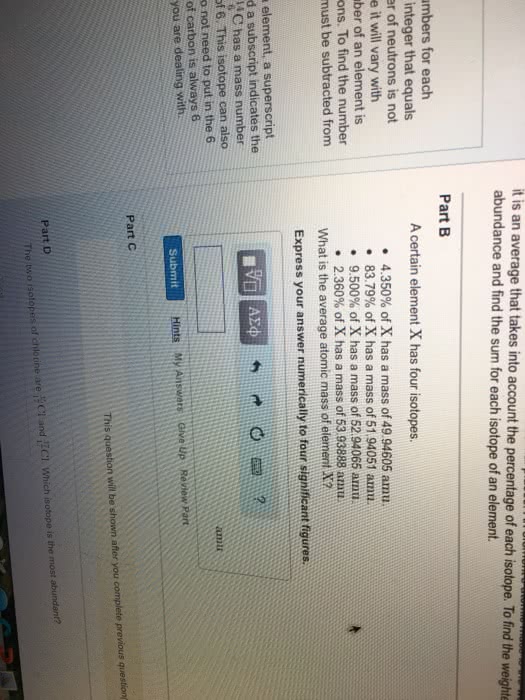
6 = atomic number = number of protons. 12 = mass number = number of protons and number of neutrons. 5 = electrons = number of protons - charge. No charge in this example, protons = electrons = 26. Isotopes: atoms with the same number of protons but different number of neutrons. 20 ne can also be written neon- 20. Average mass: mass numbers are always whole numbers but the mass on the periodic table is a decimal. Mass found on periodic table is the average mass. Atomic mass = (fraction of isotope 1 x mass of isotope 1) + (fraction of isotope 2 x mass. Calculate it from mass number of all isotopes in natural abundance of isotope 2) + When finding the fraction of the isotope, move the decimal to places to the left (dividing by 100) How many of each atom there is, exact molecular. Simplest formula, divide everything by the smallest number, ratios empirical.