CHEM 1A Lecture Notes - Lecture 22: Trigonal Planar Molecular Geometry, Orbital Hybridisation, Sigma Bond
CHEM 1A verified notes
22/31View all
Document Summary
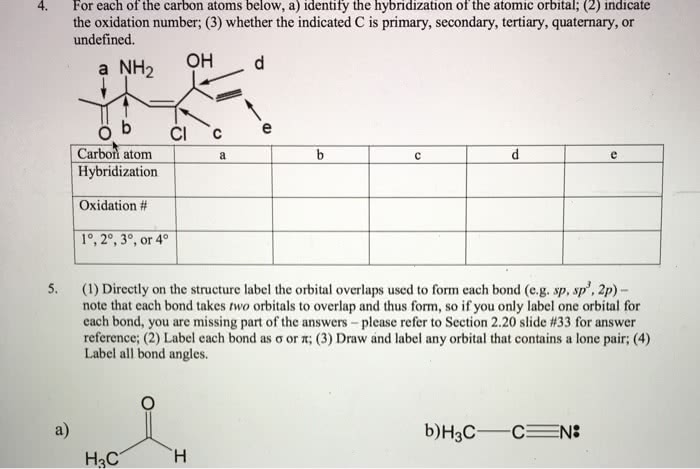
Aleks due 12/3, but do work on it to prepare for exam 2 this friday. Mixed to make 3 sp 2 hybridized orbitals on c. And the remaining p is not affected. As shown in the diagram on the top-right, the resultant orbitals are 1 p-orbital and 3 sp 2 orbitals . This is how we have a trigonal planar shape of. Recall that a direct overlap of orbitals is a sigma bond. And an indirect (sideway) overlap of orbitals is a pi bond. When 2 carbon atoms come together, one of their sp 2 orbitals will overlap first, forming the sigma bond, and then their p-orbital will overlap partially, forming the pi bond. Hence a c=c double bond has 1 sigma bond, and 1 pi bond (squeezing on top and below the carbons) Also notice h has only 1s, so it can only form sigma bond.