CHM136H1 Lecture Notes - Lecture 3: Alkane, Bromine, Ethylene
12 views10 pages

34
CHM136H1 Full Course Notes
Verified Note
34 documents
Document Summary
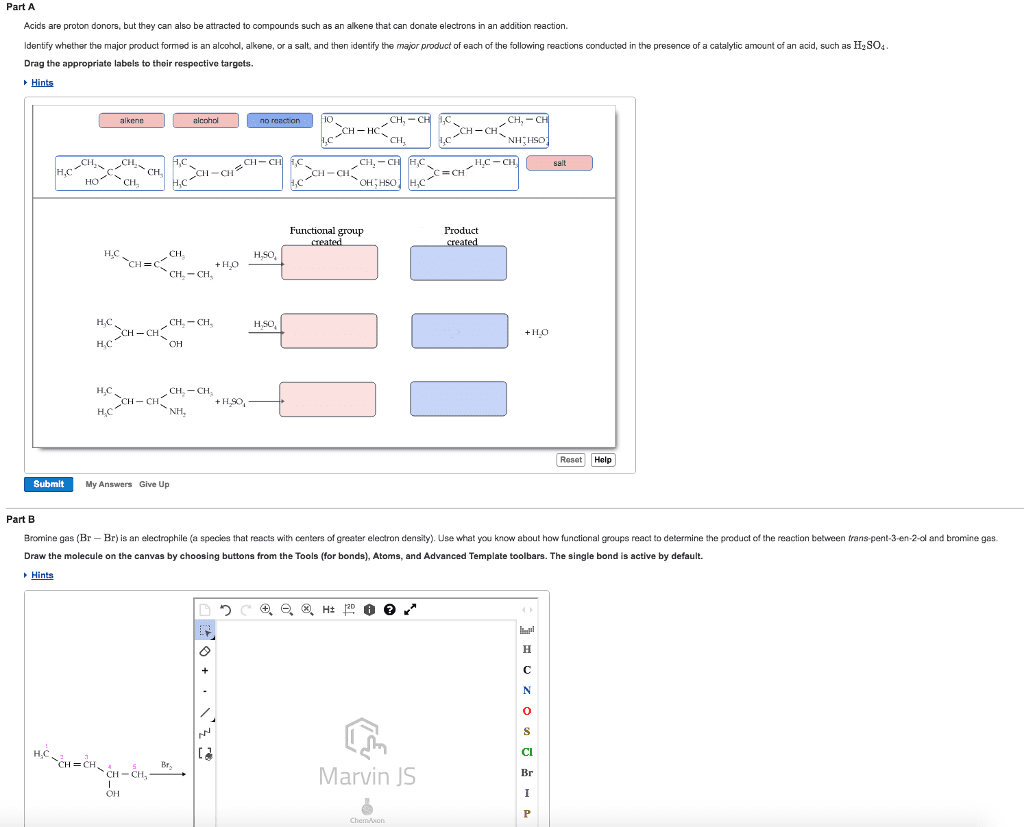
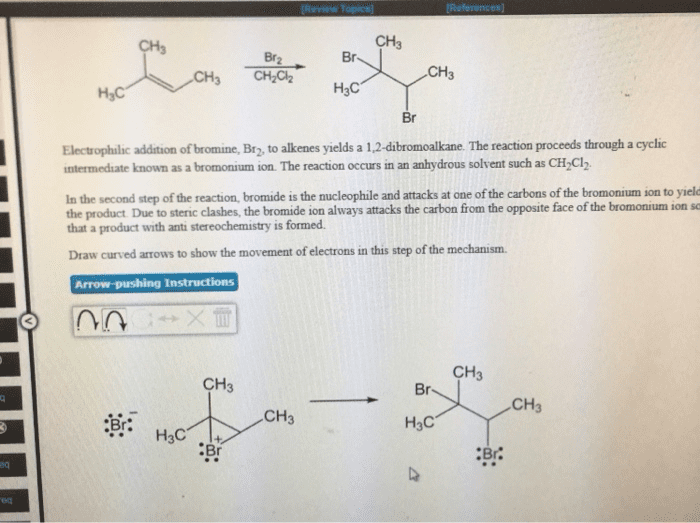
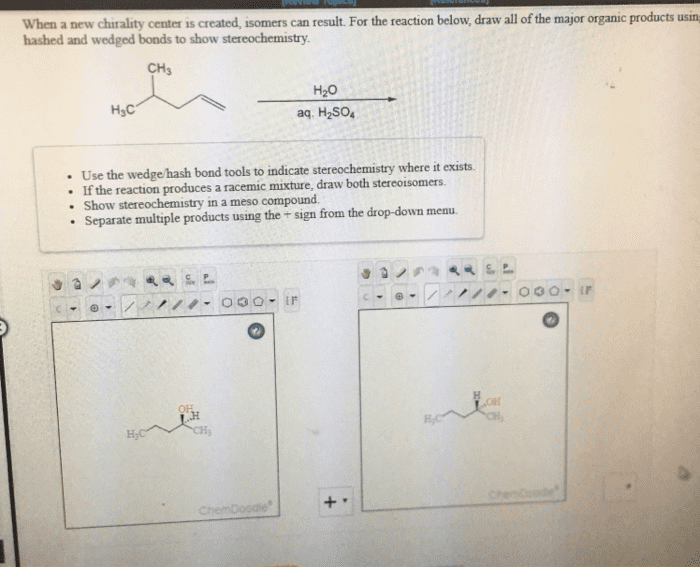
Functional group: a group of atoms having characteristic behaviour in all molecules it occurs. The group reacts predictably (usually independent to the rest of the molecule) The double bond c=c reacts identically whether it is in a simple alkene or and a complex alkene: Ethylene has a c=c: ethylene can react with bromine to add one br on each c. Menthene also has a c=c: menthene can react with bromine to add one br on each c. Cholesterol has a c=c: cholesterol can react with bromine to add one br on each c ( dibromocholesterol ) Functional groups that contain hydrogen and carbon atoms. There are 3 different kinds: alkene (double bond) (e. g. c=c, alkyne (triple bond) (e. g. c c, arene; aromatic ring (three pi bonds linked together) Bonds are not specified: only shows two carbon; not what they are attached to. The heteroatom will be more electronegative than c. Alkyl halide (haloalkanes): halogen directly bonded to c.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers