CHM136H1 Lecture 3: Alkanes & Their Stereochemistry (Lectures 7-9)

34
CHM136H1 Full Course Notes
Verified Note
34 documents
Document Summary
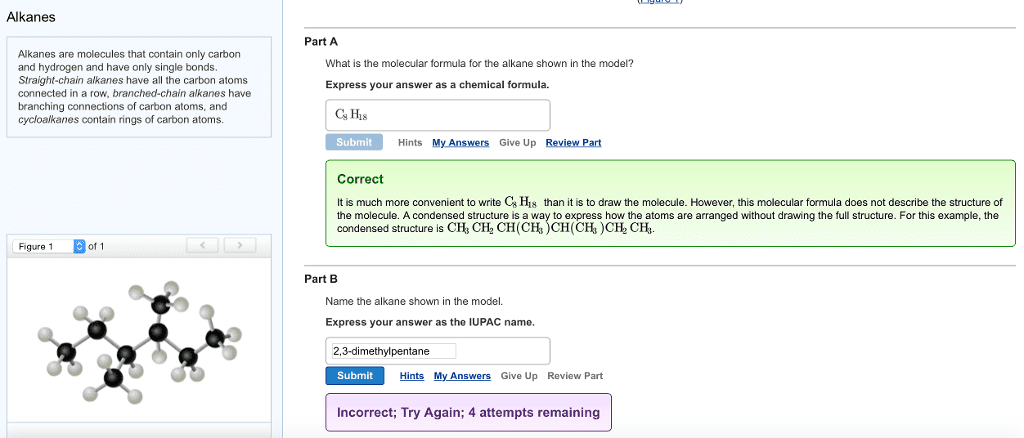
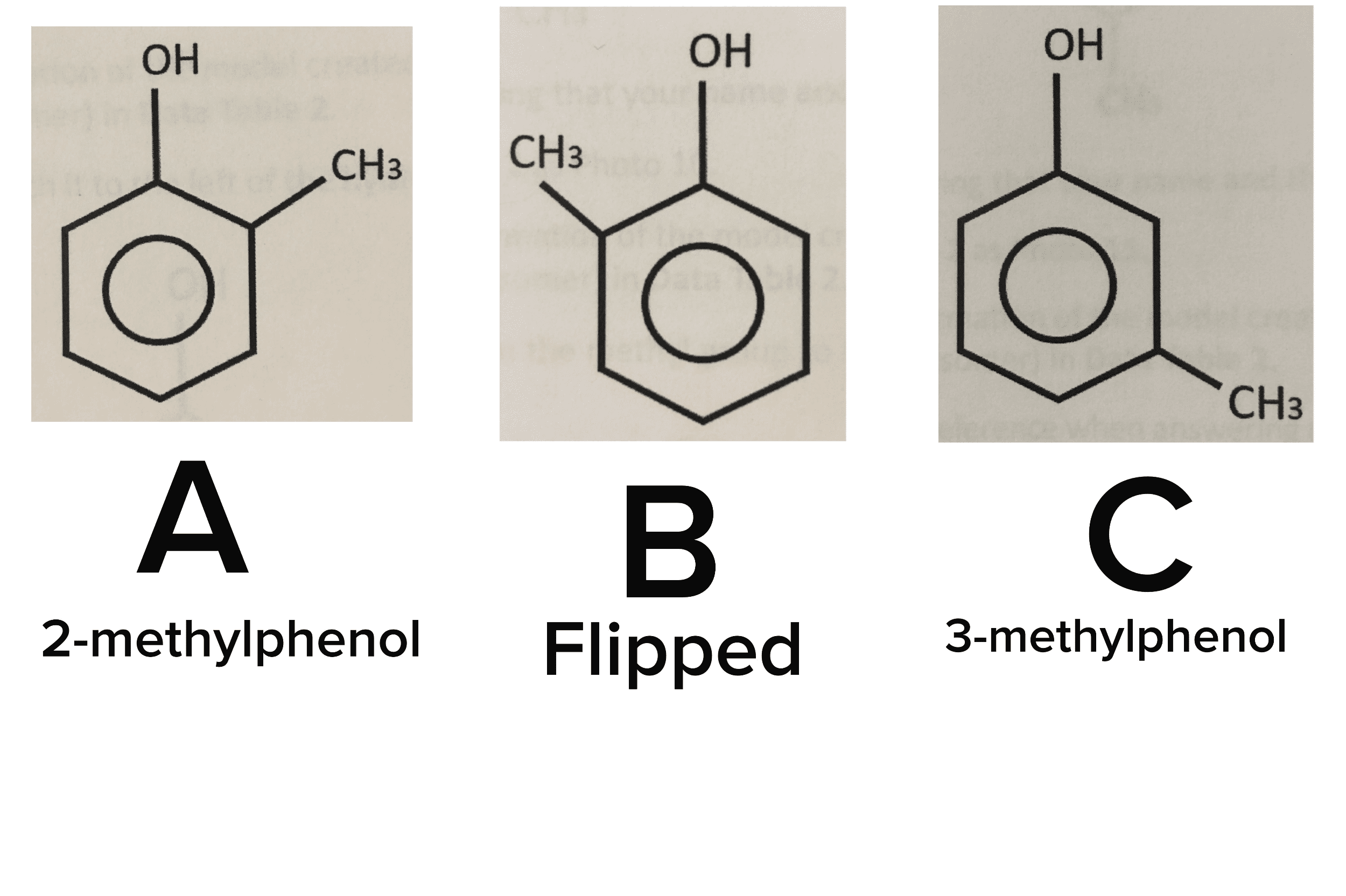
Chm 236 : alkanes & their stereochemistry alkanes : saturated hydrocarbons stereochemistry : the. 3d arrangement of atoms in a molecule of interest. Functionalgro functions : a group of atoms having characteristic behaviour in all molecules it occurs reacts predictably. Iusually independent of the rest of the molecule) Cn hzn - 2 alkyne: triple bond ; General formula : th - cen functional group nitrile. Functional group : carbonyl ht another oxygen singly bonded to it functional group : sulfide chlorine : acid. R - o - r " functional group : ahh oxy. 13 " functional group : alkyl t nitrogen atom. Throat @ atom finstead of 0 atom) sulfur. C - c single bonds and c - h bonds only. I hydrogen atoms alkanes with more than 3 carbon atoms have isomers prefixes. : any compounds differing in (cid:8869) must have the same molecular formula same formula different bonds how their atoms are connected to each other.