MBB 222 Lecture Notes - Lecture 3: Benzene, Butane, Stereoisomerism
Document Summary
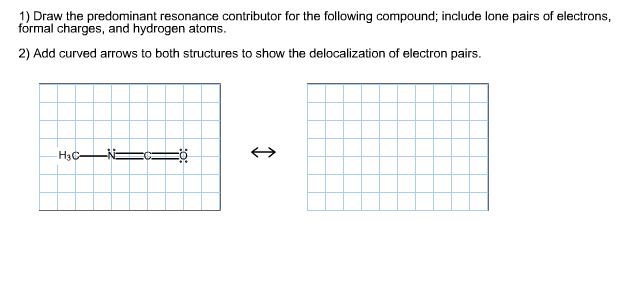
Localizedus delocalizedes ab iviti de havediscrete singleanddoublebonds cuz ab localizedes im x. Ch c s abe to resonance contributors s o case. Q. is indelocalized es resonancehybrid themoleculeabovedoesnothavediscretesingleanddouble c obonds thee pairscirculateacrosstheo c o atoms. Delocalization is animportantpropertyofproteinspeptides os n ti i ii ecu. H tidebond s ca i en ti r resonancecontributors a ch i r n l it resonancehybrid aromaticcompounds exbenzene. H i c ee s h i c c h i. H ex h c i a c h i ti. H resonancehybrid aromaticmolecule benzene vs aliphaticmoiec cyclohexane a benzene cyclohexane cyaizedocarbonringw carbons linkedbyalteringsingle doublebonds linkedbysinglebonds cyclized 6 carbonringwicarbons each c has in each c has zits rigidplanarstructure. Aliphaticmolecule flexible nonplanarstructure aromaticcompounds ie s aredelocalized in a aclouds ii resonance uniquepropertiesduetoresonance verystable i notreactive. Benzenhasdelocalized es aplanarmolecule has6identicalcarbon carbonbonds eacha e is sharedbyan6 carbons es are delocalized onlycyclicmomthatforma closedloopofoverlapping a orbitalsarearomatic aromaticcompoundshavedistinctphysicalpropertiesthataffecttheirinteractionswieachother othermore. I flatmolecularstructure a tendencytostackupononeanother esp inwater aromaticcompoundscancontain n o s heterocyclic aromaticcompounds canalsobe smemberedrings ex.