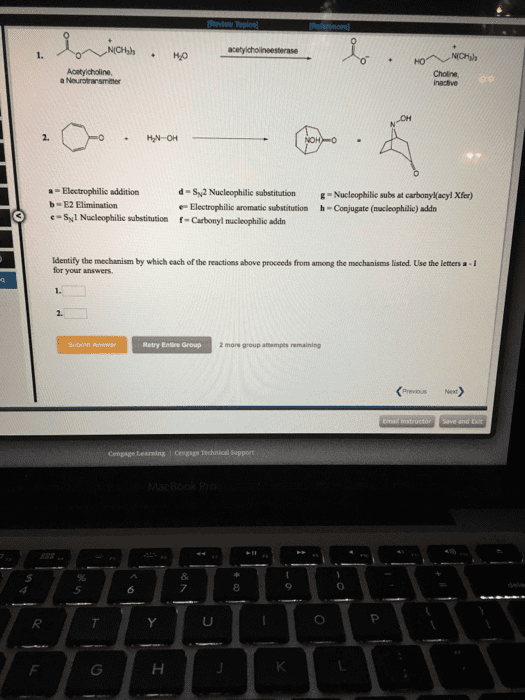
MBB 222 Lecture Notes - Lecture 19: Electrophile, Nucleophile, Enzyme
Get access


Related Documents
Related Questions
What is the first step in the mechanism of an EAS reaction?
| The formation of a strong electrophile. | |
| Loss of a proton from the arenium ion. | |
| The attenuation of the electrophilicity of an electrophile. | |
| The diminution of the nucleophilicity of the benzene's Ï-electrons. | |
| Loss of a proton from the nucleophile. | |
| Arenium ion incontinence. | |
| The formation of a strong nucleophile. | |
| Benzene's Ï-electrons nucleophilicly attacking an electrophile. |
What is the second step in the mechanism of an EAS reaction?
| The attenuation of the electrophilicity of an electrophile. | |
| The formation of a strong nucleophile. | |
| Benzene's Ï-electrons nucleophilicly attacking an electrophile. | |
| Loss of a proton from the arenium ion. | |
| The diminution of the nucleophilicity of the benzene's Ï-electrons. | |
| Arenium ion incontinence. | |
| Loss of a proton from the nucleophile. | |
| The formation of a strong electrophile. |
What is the third step in the mechanism of an EAS reaction?
| Arenium ion incontinence. | |
| The attenuation of the electrophilicity of an electrophile. | |
| The diminution of the nucleophilicity of the benzene's Ï-electrons. | |
| Loss of a proton from the nucleophile. | |
| Benzene's Ï-electrons nucleophilicly attacking an electrophile. | |
| Loss of a proton from the arenium ion. | |
| The formation of a strong electrophile. | |
| The formation of a strong nucleophile. |