2
answers
0
watching
766
views
26 Sep 2018
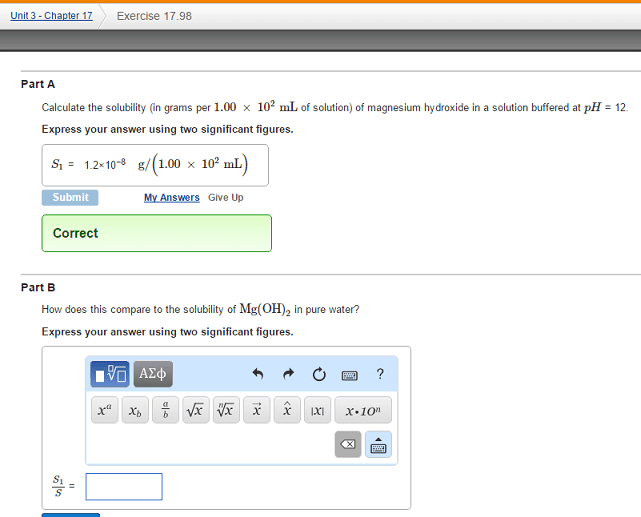
98. Calculate the solubility (in grams per 1.00 X 102 mL of solu- tion) of magnesium hydroxide in a solution buffered at pH = 10. How does this compare to the solubility of Mg(OH)2 in pure water?
98. Calculate the solubility (in grams per 1.00 X 102 mL of solu- tion) of magnesium hydroxide in a solution buffered at pH = 10. How does this compare to the solubility of Mg(OH)2 in pure water?
Elin HesselLv2
28 Sep 2018
Already have an account? Log in