0
answers
0
watching
73
views
11 Dec 2019
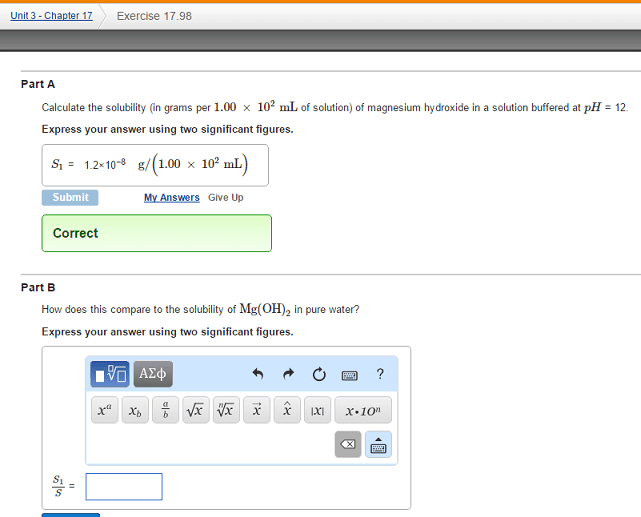
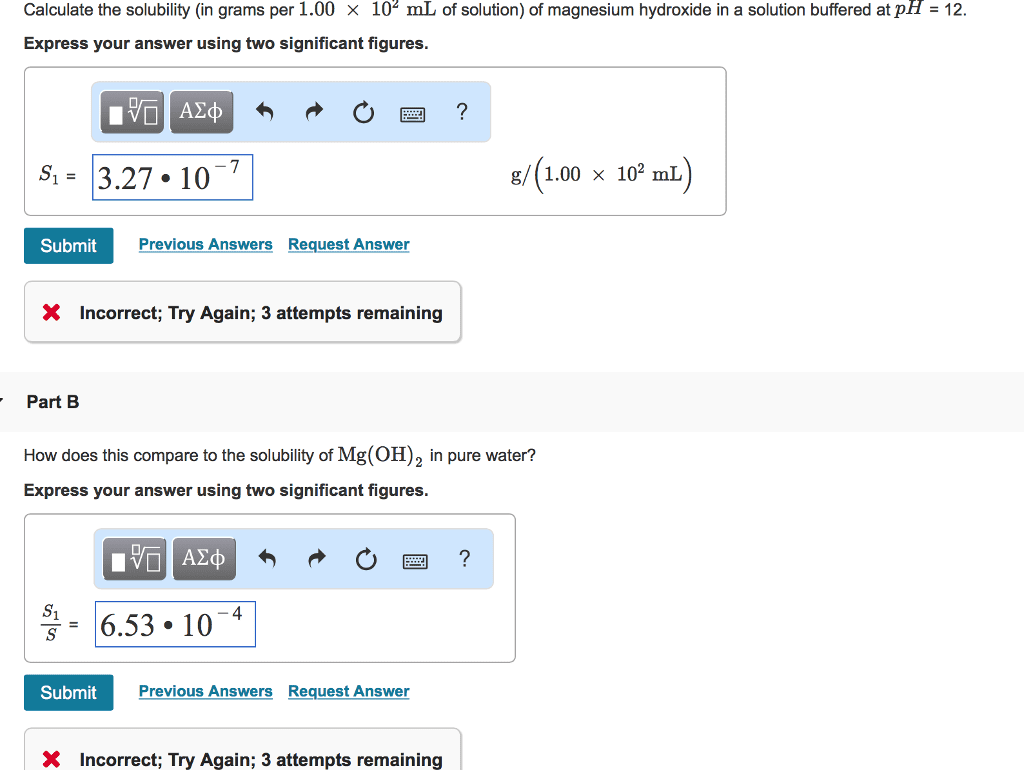
A)Calculate the solubility (in grams per 1.00Ã102mL of solution) of magnesium hydroxide in a solution buffered at pH = 12.(answer is S1 = 1.2Ã10â8 g/(1.00Ã102mL) B)How does this compare to the solubility of Mg(OH)2 in pure water?
A)Calculate the solubility (in grams per 1.00Ã102mL of solution) of magnesium hydroxide in a solution buffered at pH = 12.(answer is S1 = 1.2Ã10â8 g/(1.00Ã102mL) B)How does this compare to the solubility of Mg(OH)2 in pure water?