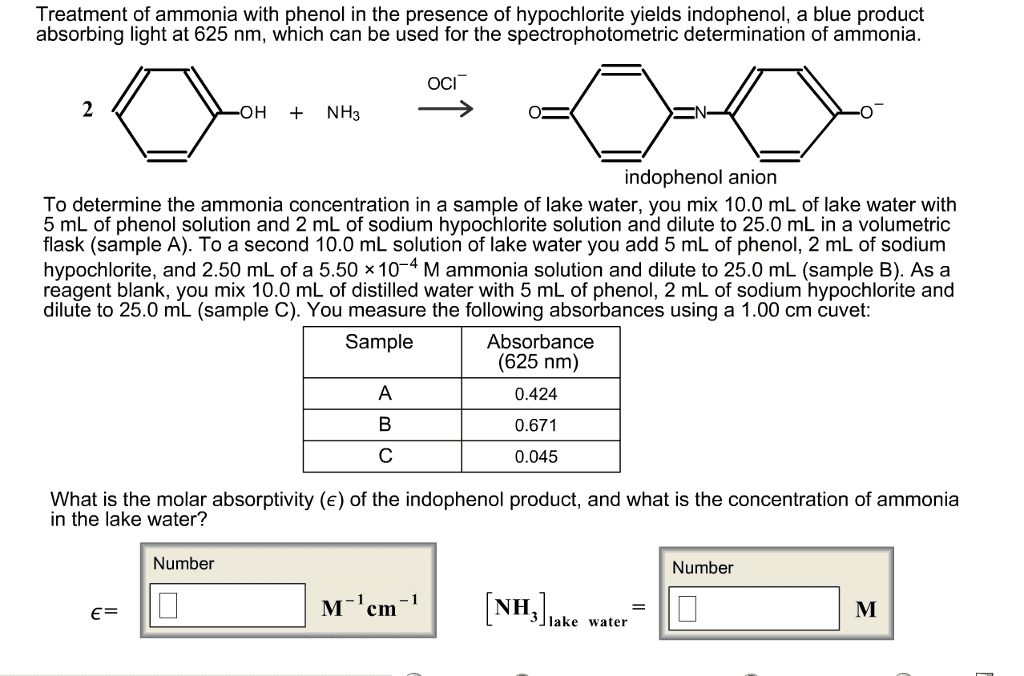
Treatment of ammonia with phenol in the presence of hypochloriteyields indophenol, a blue product absorbing light at 625 nm, whichcan be used for the spectrophotometric determination ofammonia.
To determine the ammonia concentration in a sample of lakewater, you mix 10.0 mL of lake water with 5 mL of phenol solutionand 2 mL of sodium hypochlorite solution and dilute to 25.0 mL in avolumetric flask (sample A). To a second 10.0 mL solution of lakewater you add 5 mL of phenol, 2 mL of sodium hypochlorite, and 2.50mL of a 5.50
Treatment of ammonia with phenol in the presence of hypochloriteyields indophenol, a blue product absorbing light at 625 nm, whichcan be used for the spectrophotometric determination ofammonia.
To determine the ammonia concentration in a sample of lakewater, you mix 10.0 mL of lake water with 5 mL of phenol solutionand 2 mL of sodium hypochlorite solution and dilute to 25.0 mL in avolumetric flask (sample A). To a second 10.0 mL solution of lakewater you add 5 mL of phenol, 2 mL of sodium hypochlorite, and 2.50mL of a 5.50
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
Treatment of ammonia with phenol in the presence of hypochlorite yields indophenol, a blue product absorbing light at 625 nm, which can be used for the spectrophotometric determination of ammonia.
To determine the ammonia concentration in a sample of lake water, you mix 10.0 mL of lake water with 5 mL of phenol solution and 2 mL of sodium hypochlorite solution and dilute to 25.0 mL in a volumetric flask (sample A). To a second 10.0 mL solution of lake water you add 5 mL of phenol, 2 mL of sodium hypochlorite, and 2.50 mL of a 5.50 Ã 10â4 M ammonia solution and dilute to 25.0 mL (sample B). As a reagent blank, you mix 10.0 mL of distilled water with 5 mL of phenol, 2 mL of sodium hypochlorite and dilute to 25.0 mL (sample C). You measure the following absorbances using a 1.00 cm cuvet:
| Sample | Absorbance(625 nm) |
| A | .385 |
| B | .632 |
| C | .045 |
What is the molar absorptivity (ε) of the indophenol product, and what is the concentration of ammonia in the lake water?