1
answer
0
watching
270
views
10 Nov 2019
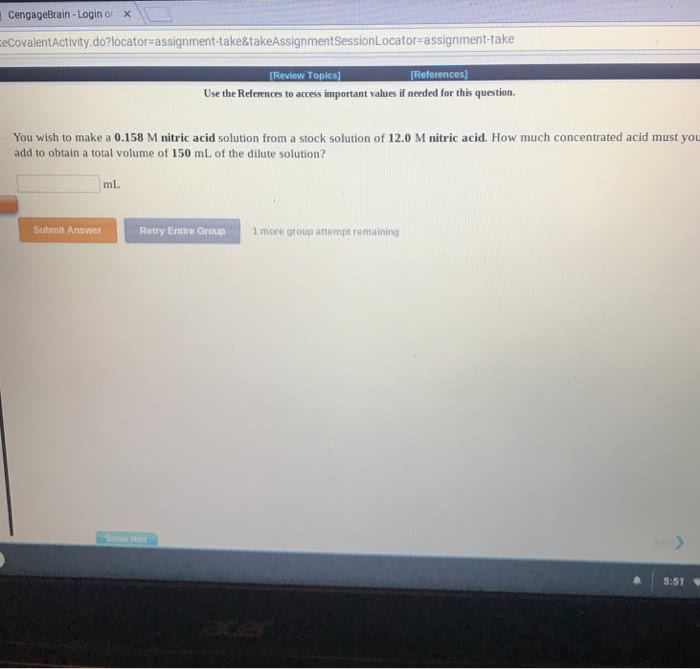
You wish to make a 0.286 M hydroiodic acid solution from a stocksolution of 12.0 M hydroiodic acid. How much concentrated acid mustyou add to obtain a total volume of 75.0 mL of the dilutesolution?
You wish to make a 0.286 M hydroiodic acid solution from a stocksolution of 12.0 M hydroiodic acid. How much concentrated acid mustyou add to obtain a total volume of 75.0 mL of the dilutesolution?
Patrina SchowalterLv2
10 Nov 2019