1
answer
0
watching
85
views
11 Nov 2019
1 and 2
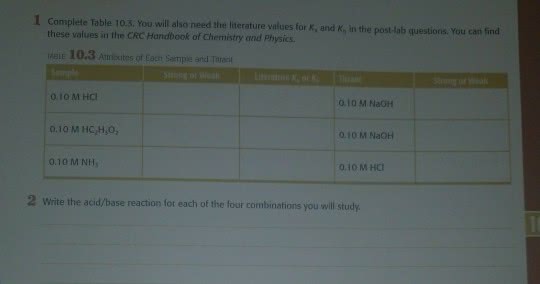
1 Complete Table 10.3. You will alsto need the literature values for K, and K, in the post Jab questions. You can find these values in the CRC Handbook of Chemistry and Physics. TABLE 10.3 Attributes of Each Sample and Tarant ong or 0.10 M HC 0.10 M NaOH 0.10 M HC H.O, 0.10 M NaOH 0.10 M NH, 0.10 M HCI 2 write the acid/base reaction for each of the four combinations you will study
1 and 2

1 Complete Table 10.3. You will alsto need the literature values for K, and K, in the post Jab questions. You can find these values in the CRC Handbook of Chemistry and Physics. TABLE 10.3 Attributes of Each Sample and Tarant ong or 0.10 M HC 0.10 M NaOH 0.10 M HC H.O, 0.10 M NaOH 0.10 M NH, 0.10 M HCI 2 write the acid/base reaction for each of the four combinations you will study
1
answer
0
watching
85
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Sixta KovacekLv2
30 Oct 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232