1
answer
0
watching
1,126
views
28 Sep 2019
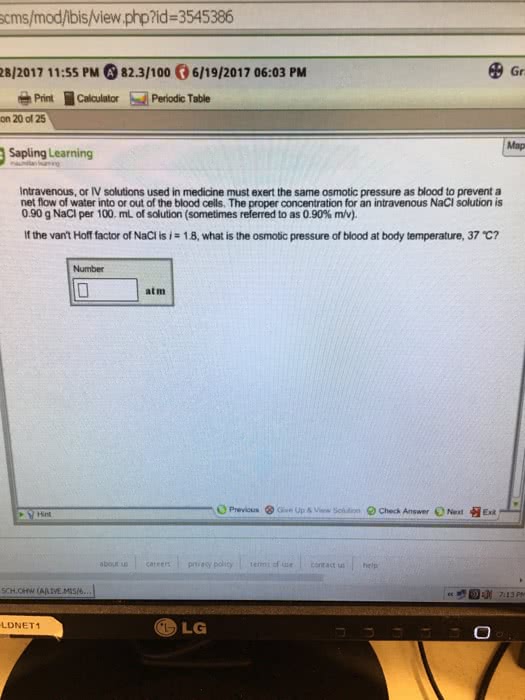
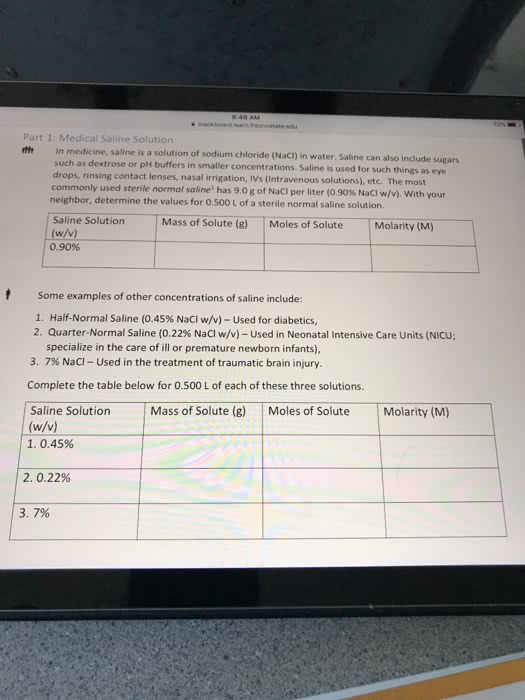
Intravenous, or IV solutions used in medicine must exert the same osmotic pressure as blood to prevent a net flow of water into or out of the blood cells. The proper concentration for an intravenous NaCl solution is 0.90 g NaCl per 100 mL of water (sometimes referred to as 0.90% m/v).If the van
Intravenous, or IV solutions used in medicine must exert the same osmotic pressure as blood to prevent a net flow of water into or out of the blood cells. The proper concentration for an intravenous NaCl solution is 0.90 g NaCl per 100 mL of water (sometimes referred to as 0.90% m/v).If the van
Jean KeelingLv2
28 Sep 2019