1
answer
0
watching
341
views
23 Nov 2019
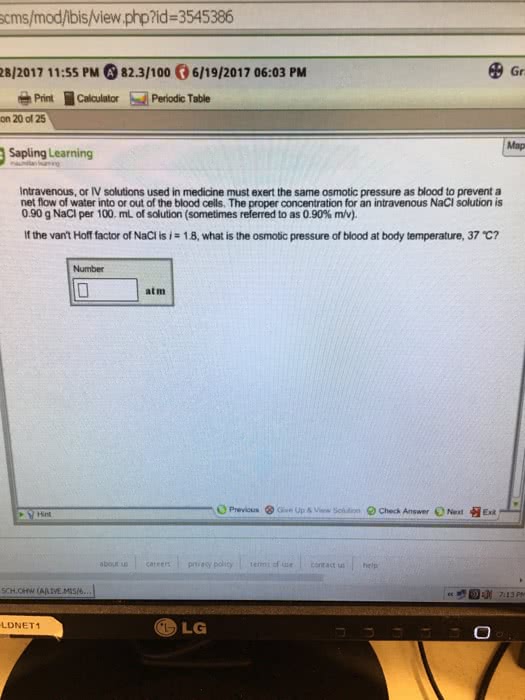
Intravenous, or IV solutions used in medicine must exert the same osmotic pressure as blood to prevent a net flow of water into or out of the blood cells. The proper concentration for an intravenous NaCl solution is 0.90 g NaCl per 100. mL of water (sometimes refered to as 0.90% m/v). If the van\'t Hoff factor of NaCl is i = 1.8, what is the osmotic pressure of blood at body temperatrue, 37
Intravenous, or IV solutions used in medicine must exert the same osmotic pressure as blood to prevent a net flow of water into or out of the blood cells. The proper concentration for an intravenous NaCl solution is 0.90 g NaCl per 100. mL of water (sometimes refered to as 0.90% m/v). If the van\'t Hoff factor of NaCl is i = 1.8, what is the osmotic pressure of blood at body temperatrue, 37
Jamar FerryLv2
19 Aug 2019