1
answer
0
watching
124
views
16 Nov 2019
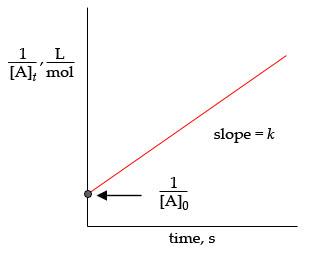
Question 5: Given the reaction data below, plot the data assuming both a first order and second order reaction. If a plot of ln[A] vs. time is linear, the reaction is first order (unimolecular). If a plot of 1/[A] vs. time is linear, the reaction is second order (bimolecular). This will be very fast if you use a spreadsheet program, like Excel. a) What is the molecularity of this reaction? b) Calculate the rate constant (this will be the slope of the straight line). Time (s) 0,1,2,3,4,5,Reactant mM 6.2,3.1,2.1,1.6,1.3,1.1
Question 5: Given the reaction data below, plot the data assuming both a first order and second order reaction. If a plot of ln[A] vs. time is linear, the reaction is first order (unimolecular). If a plot of 1/[A] vs. time is linear, the reaction is second order (bimolecular). This will be very fast if you use a spreadsheet program, like Excel. a) What is the molecularity of this reaction? b) Calculate the rate constant (this will be the slope of the straight line). Time (s) 0,1,2,3,4,5,Reactant mM 6.2,3.1,2.1,1.6,1.3,1.1
Jarrod RobelLv2
2 Sep 2019