1
answer
0
watching
83
views
23 Nov 2019
State whether the following are characteristics of zero-, first-, or second-order kinetics for the reaction R ----> P. If there is not enough information given to determine the order of the reaction, indicate so.
Use the following key to indicate your answer:
0 = zero order 1 = first order 2 = second order N/A = insufficient information
(a) ___ The reaction rate is not affected by [R].
(b) ___ A plot of ln[R] vs. time is linear.
(c) ___ The reaction rate increases with increasing temperature.
(d) ___ A catalyst can accelerate the reaction.
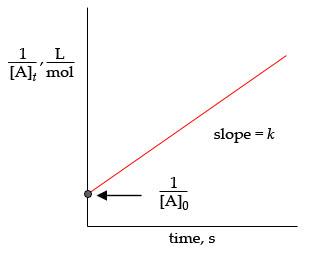
(e) ___ A plot of 1/[R] vs. time is linear.
State whether the following are characteristics of zero-, first-, or second-order kinetics for the reaction R ----> P. If there is not enough information given to determine the order of the reaction, indicate so.
Use the following key to indicate your answer:
0 = zero order 1 = first order 2 = second order N/A = insufficient information
(a) ___ The reaction rate is not affected by [R].
(b) ___ A plot of ln[R] vs. time is linear.
(c) ___ The reaction rate increases with increasing temperature.
(d) ___ A catalyst can accelerate the reaction.
(e) ___ A plot of 1/[R] vs. time is linear.
Elin HesselLv2
27 Jul 2019