1
answer
0
watching
261
views
16 Nov 2019
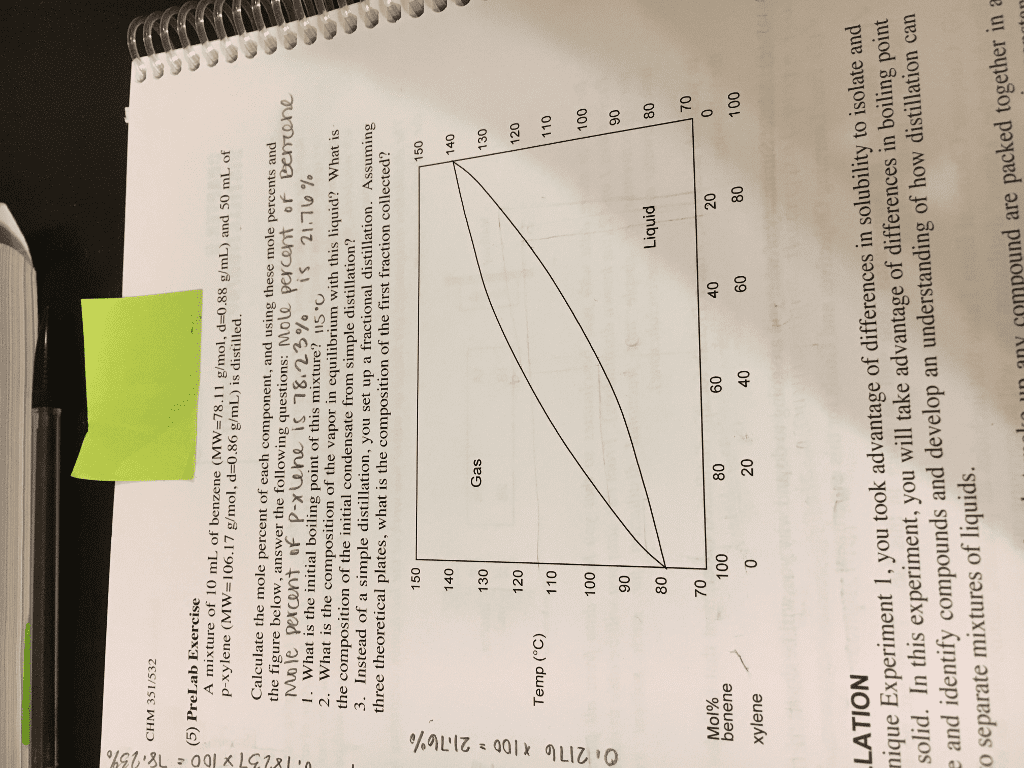
Let's push a little further. You are using simple distillation to purify your mixture of 40 mole % Compound A and 60 mole % Compound B and want to purify your sample to be at least 90% Compound A. What is the smallest number of distillations you need to perform to achieve at least 90% Compound A in your distillate? (2 pts)

80 70 oc 60 50 20 40 60 80 100 mole B
Let's push a little further. You are using simple distillation to purify your mixture of 40 mole % Compound A and 60 mole % Compound B and want to purify your sample to be at least 90% Compound A. What is the smallest number of distillations you need to perform to achieve at least 90% Compound A in your distillate? (2 pts)

80 70 oc 60 50 20 40 60 80 100 mole B
1
answer
0
watching
261
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Nestor RutherfordLv2
8 Nov 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232