1
answer
0
watching
177
views
17 Nov 2019
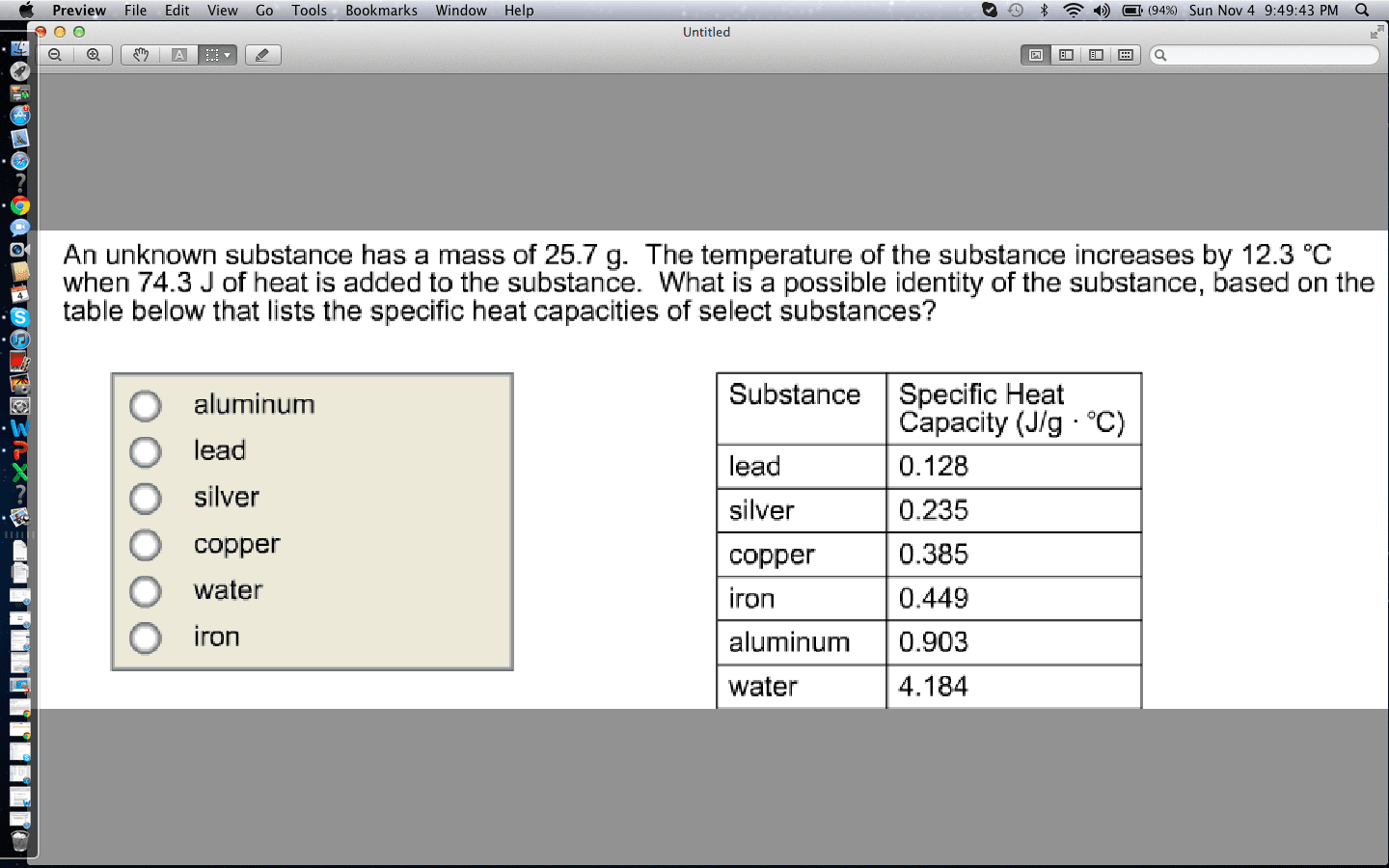
Suppose you have an equal mass of each of these metals, which one will experience the greatest change in temperature if 1000 joules of energy is added? Explain your reasoning.
Substance Specific Heat Capacity Aluminum 0.903 J/(g·°C) Copper 0.385 J/(g·°C) Gold 0.128 J/(g·°C) Iron 0.449 J/(g·°C) Lead 0.128 J/(g·°C) Silver 0.235 J/(g·°C) Ethanol 2.42 J/(g·°C) Water 4.184 J/(g·°C) Propylene glycol 2.52 J/(g·°C)
Suppose you have an equal mass of each of these metals, which one will experience the greatest change in temperature if 1000 joules of energy is added? Explain your reasoning.
| Substance | Specific Heat Capacity |
| Aluminum | 0.903 J/(g·°C) |
| Copper | 0.385 J/(g·°C) |
| Gold | 0.128 J/(g·°C) |
| Iron | 0.449 J/(g·°C) |
| Lead | 0.128 J/(g·°C) |
| Silver | 0.235 J/(g·°C) |
| Ethanol | 2.42 J/(g·°C) |
| Water | 4.184 J/(g·°C) |
| Propylene glycol | 2.52 J/(g·°C) |
Irving HeathcoteLv2
5 Apr 2019