Procedure Part A:
Oxone reaction: Dissolve 0.40 g Oxone ® in 2.0 mL of water by
shaking vigorously, but carefully, in a sealed vial. (SAFETY: Be sure that the flask is
sealed well. Oxone ® solution is strongly oxidizing and should not come into contact
with skin.) Dissolve 50 ?L of cyclohexene in 2.0 mL of acetone in a small Erlenmeyer
flask. Cool the cyclohexene solution in an icebath, then add the oxone solution dropwise
over 5 minutes with swirling in the icebath. Remove the Erlenmeyer flask from the
icebath and allow the reaction to sit for at least 15 minutes. (This is a good point to begin
the permanganate reaction.) After 15-30 minutes, add about 0.1 mL of conc. HCl to the
reaction dropwise with swirling. Allow the reaction to sit for about 10 minutes, swirling
occasionally.
Procedure Part B:
Potassium permanganate reaction: Dissolve 100 mg of KMnO4 in
4.0 mL of 0.1 M NaOH solution in a small Erlenmeyer flask. Cool the KMnO4 solution
in an icebath. Dissolve 50 ?L of cyclohexene in 2.0 mL of t-butanol in a separate
container. Quickly add the cyclohexene solution into the potassium permanganate. Swirl
the flask in the icebath for 3-5 minutes. Remove the Erlenmeyer flask from the icebath
and allow to sit for 10 minutes.
Procedure Part C:
Thin Layer Chromatography: Obtain a TLC plate. Mark the plate
with four marks, two for each reaction mixture. Spot the crude reaction mixtures lightly.
It may be hard to spot the KMnO4 slurry, but keep trying, you may find it easier if you
use a slightly wider capillary for this one. Allow the plate to dry for a couple of minutes,
or use a heat gun to gently dry the aqueous spots. Develop the plate in a chamber with
ethyl acetate as the eluent. Remove it from the chamber and mark the solvent front. Stain
the plate using anisaldehyde stain, and heat the plate gently to develop the spots. The cis
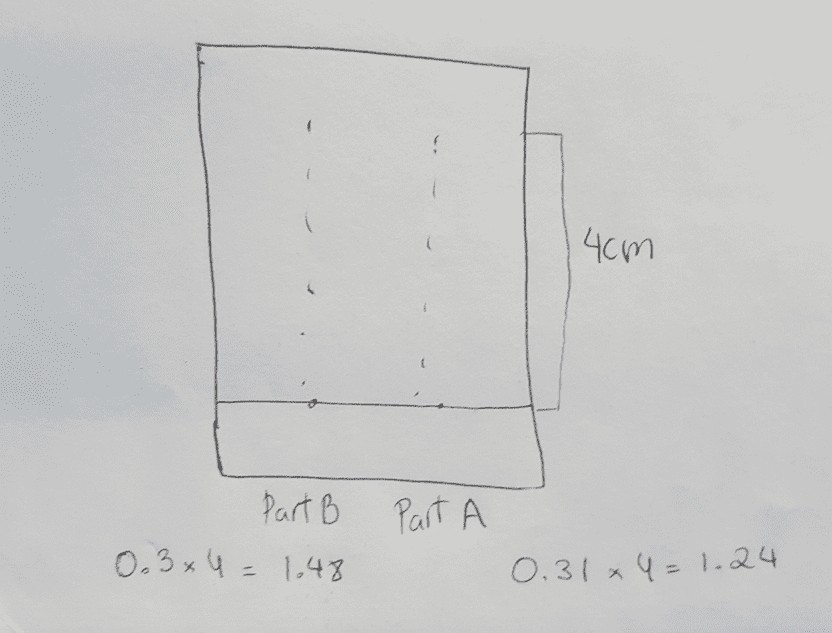
isomer should stain red, and have an Rf value of ~0.37, the trans isomer should stain blue,
and have an Rf value of ~0.31.
Questions
1. Draw a sketch of the TLC analysis for both reactions.
2. Calculate the Rf of both products formed.
3. Using you text and any other information you can find predict the products of the
4. reactions you performed.
5. Are these reactions stereoselective, stereopsecific or do they give mixtures?
Part A O. 3 1.24 l 48
Procedure Part A:
Oxone reaction: Dissolve 0.40 g Oxone ® in 2.0 mL of water by
shaking vigorously, but carefully, in a sealed vial. (SAFETY: Be sure that the flask is
sealed well. Oxone ® solution is strongly oxidizing and should not come into contact
with skin.) Dissolve 50 ?L of cyclohexene in 2.0 mL of acetone in a small Erlenmeyer
flask. Cool the cyclohexene solution in an icebath, then add the oxone solution dropwise
over 5 minutes with swirling in the icebath. Remove the Erlenmeyer flask from the
icebath and allow the reaction to sit for at least 15 minutes. (This is a good point to begin
the permanganate reaction.) After 15-30 minutes, add about 0.1 mL of conc. HCl to the
reaction dropwise with swirling. Allow the reaction to sit for about 10 minutes, swirling
occasionally.
Procedure Part B:
Potassium permanganate reaction: Dissolve 100 mg of KMnO4 in
4.0 mL of 0.1 M NaOH solution in a small Erlenmeyer flask. Cool the KMnO4 solution
in an icebath. Dissolve 50 ?L of cyclohexene in 2.0 mL of t-butanol in a separate
container. Quickly add the cyclohexene solution into the potassium permanganate. Swirl
the flask in the icebath for 3-5 minutes. Remove the Erlenmeyer flask from the icebath
and allow to sit for 10 minutes.
Procedure Part C:
Thin Layer Chromatography: Obtain a TLC plate. Mark the plate
with four marks, two for each reaction mixture. Spot the crude reaction mixtures lightly.
It may be hard to spot the KMnO4 slurry, but keep trying, you may find it easier if you
use a slightly wider capillary for this one. Allow the plate to dry for a couple of minutes,
or use a heat gun to gently dry the aqueous spots. Develop the plate in a chamber with
ethyl acetate as the eluent. Remove it from the chamber and mark the solvent front. Stain
the plate using anisaldehyde stain, and heat the plate gently to develop the spots. The cis
isomer should stain red, and have an Rf value of ~0.37, the trans isomer should stain blue,
and have an Rf value of ~0.31.

Questions
1. Draw a sketch of the TLC analysis for both reactions.
2. Calculate the Rf of both products formed.
3. Using you text and any other information you can find predict the products of the
4. reactions you performed.
5. Are these reactions stereoselective, stereopsecific or do they give mixtures?

