1
answer
0
watching
377
views
18 Nov 2019
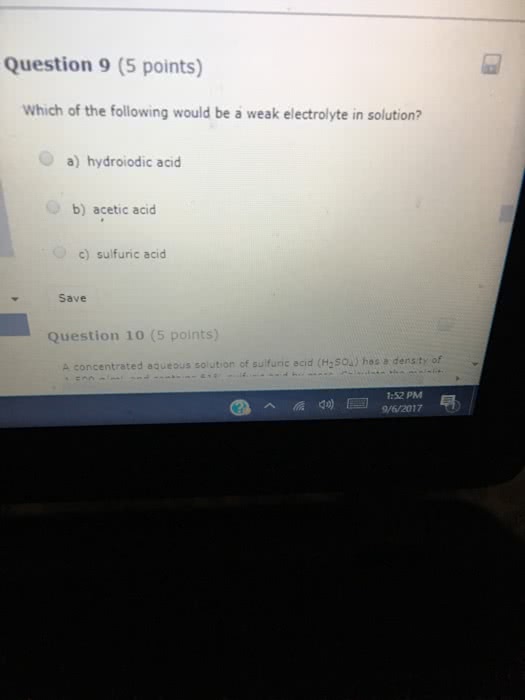
Classify each of the following as a Strong acid (sa) or a Weak acid (wa) and indicate how each should be written in aqueous solution.
1. hydroiodic acid
2. acetic acid
3. carbonic acid
Classify each of the following as a Strong acid (sa) or a Weak acid (wa) and indicate how each should be written in aqueous solution.
1. hydroiodic acid
2. acetic acid
3. carbonic acid
Beverley SmithLv2
21 Apr 2019