1
answer
0
watching
1,874
views
18 Nov 2019
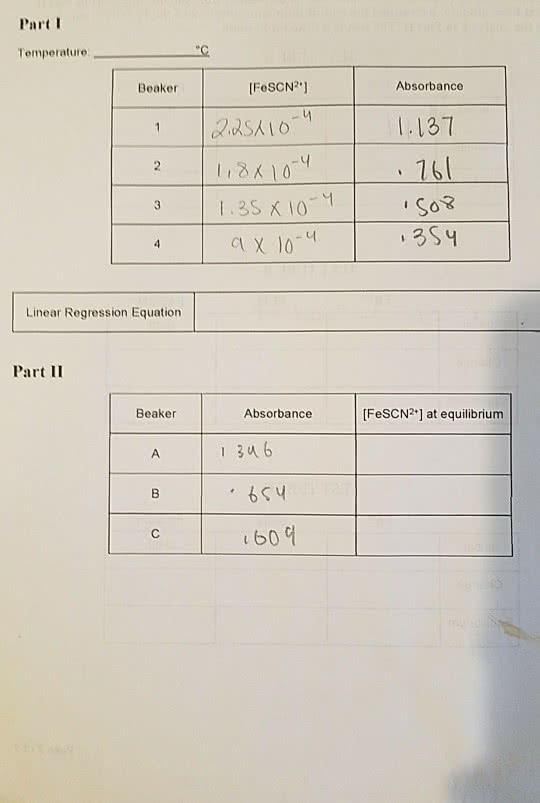
How do you calculate the initial concentration of Fe3+ and SCN-?
The ultimate goal is to find the equilbirum constant for this reaction: Fe3+ (aq) + SCN- (aq) <--> FeSCN2+ (aq)
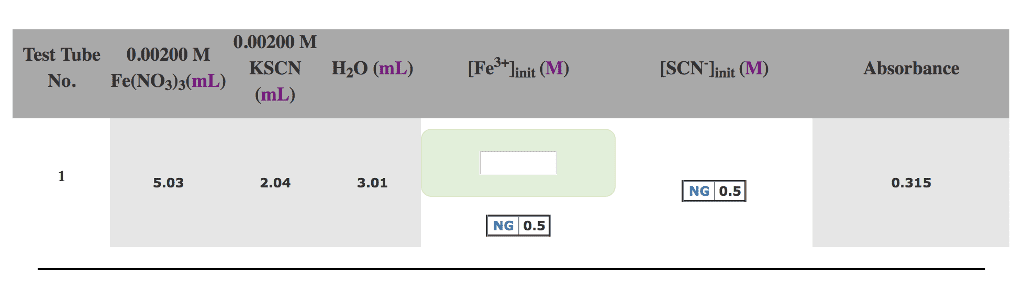
0.00200 M Test Tube 0.00200 M KSCN H20 (mL) [Fe+linit (M) [SCN linit (M) Absorbance No. Fe(NO3)3(mL) (mL) 5.03 2.04 3.01 0.315 NG 0.5 NG 0.5
How do you calculate the initial concentration of Fe3+ and SCN-?
The ultimate goal is to find the equilbirum constant for this reaction: Fe3+ (aq) + SCN- (aq) <--> FeSCN2+ (aq)

0.00200 M Test Tube 0.00200 M KSCN H20 (mL) [Fe+linit (M) [SCN linit (M) Absorbance No. Fe(NO3)3(mL) (mL) 5.03 2.04 3.01 0.315 NG 0.5 NG 0.5
Jamar FerryLv2
11 Jan 2019