1
answer
0
watching
128
views
19 Nov 2019
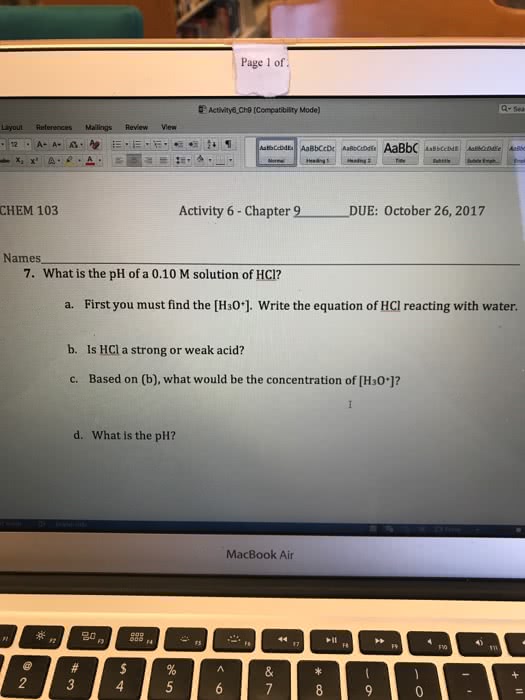

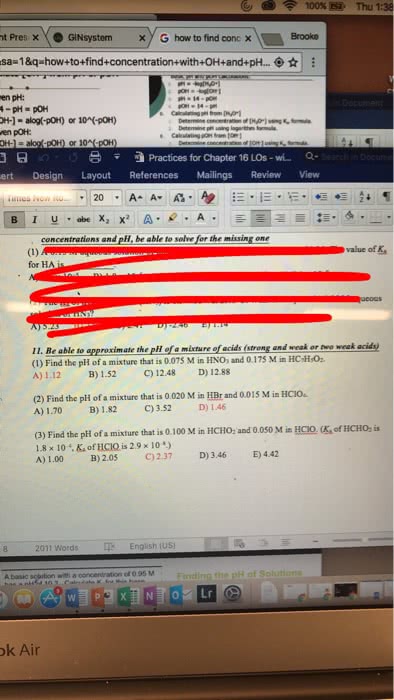
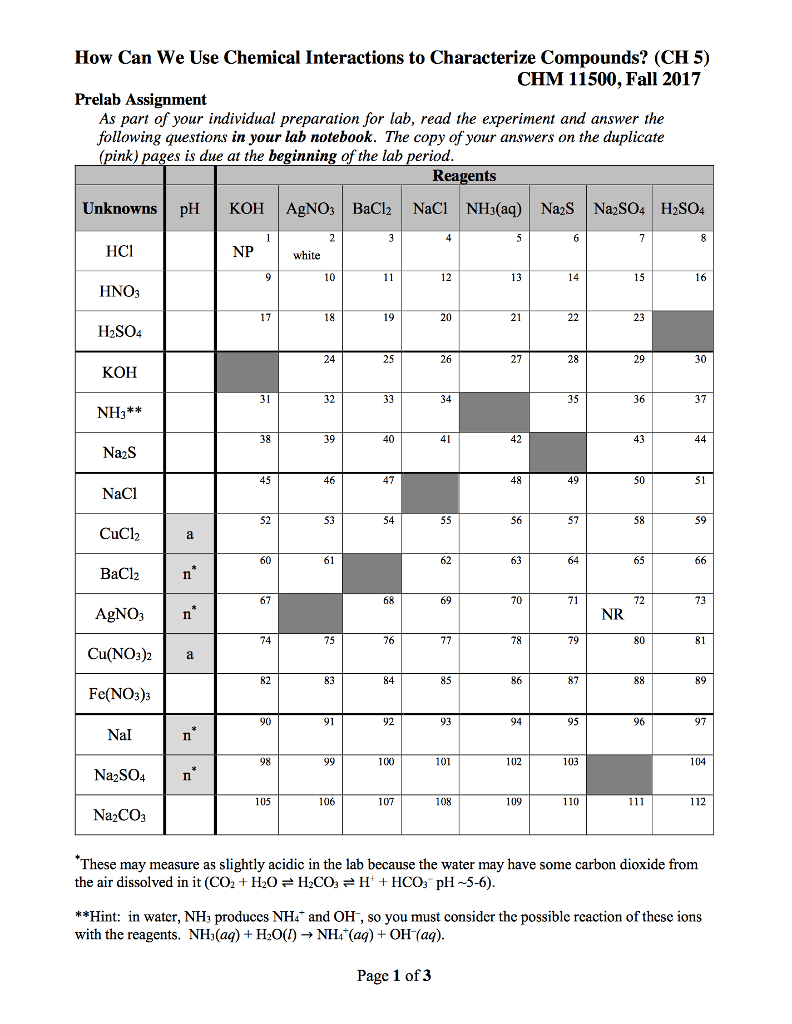
Page 1 of Q- Sea Layout References Mailings Revew View CHEM 103 Activity 6-Chapter 9 DUE: October 26, 2017 Names 7. What is the pH of a 0.10 M solution of HCI? a. First you must find the [H30 write the equation of HCl reacting with water. b. Is HCl a strong or weak acid? c. Based on (b), what would be the concentration of [H30]? d. What is the pH? MacBook Air 2 4
1
answer
0
watching
128
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Reid WolffLv2
25 Jun 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232