1
answer
0
watching
297
views
19 Nov 2019


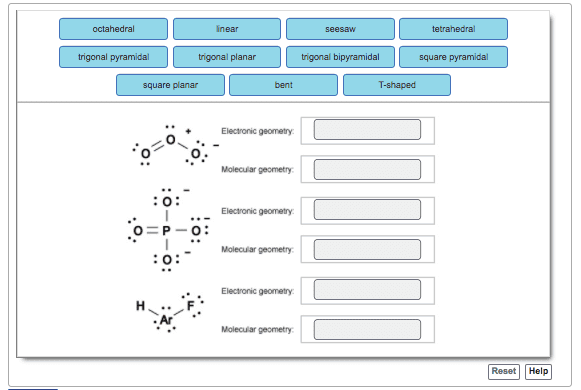
Draw the Lewis structure for NO_3^-. What are the molecular and electron geometries? Does this molecule have resonance (Circle your answer)? (a) Structure: NO_3^- (b) Molecular geometry (c) Electron geometry d) Resonance? YES/NO
1
answer
0
watching
297
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Tod ThielLv2
20 Aug 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232