In the following reactionCH4+CH4 arrow CH3CH3+H2two C-H bonds are broken(bond energy =412 kj/mol) to form a new C-C bond(bond energy=348 kj/mol and a new H-H bond(bond energy=436 kj/mol). Estimate the enthalpy in this reaction. Is it endothermic or exothermic:Hint for exothermic reactions the combined energies of the new bonds should be larger than the combined energies of the broken bonds.Balanced the following unbalanced reaction and calculate its standard free energy change(under standard reactions). State whether the reaction is spontaneous or nonspontaneous under standard conditionsC2H5OH(l)+O2(g) arrow CO2(g)+H2O(l) C2H5OH(l) O2(g) CO2(g) H2O(l) deltaH kj/mol -277.7 0 -393.5 -285.8 deltaG kjmol -174.8 0 -394.4 -237.1
In the following reactionCH4+CH4 arrow CH3CH3+H2two C-H bonds are broken(bond energy =412 kj/mol) to form a new C-C bond(bond energy=348 kj/mol and a new H-H bond(bond energy=436 kj/mol). Estimate the enthalpy in this reaction. Is it endothermic or exothermic:Hint for exothermic reactions the combined energies of the new bonds should be larger than the combined energies of the broken bonds.Balanced the following unbalanced reaction and calculate its standard free energy change(under standard reactions). State whether the reaction is spontaneous or nonspontaneous under standard conditionsC2H5OH(l)+O2(g) arrow CO2(g)+H2O(l) C2H5OH(l) O2(g) CO2(g) H2O(l) deltaH kj/mol -277.7 0 -393.5 -285.8 deltaG kjmol -174.8 0 -394.4 -237.1
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
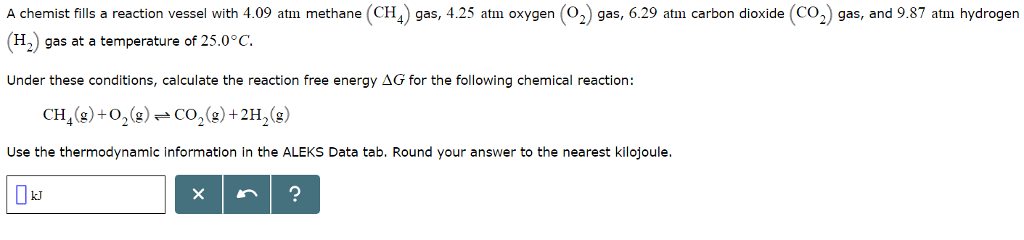
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

