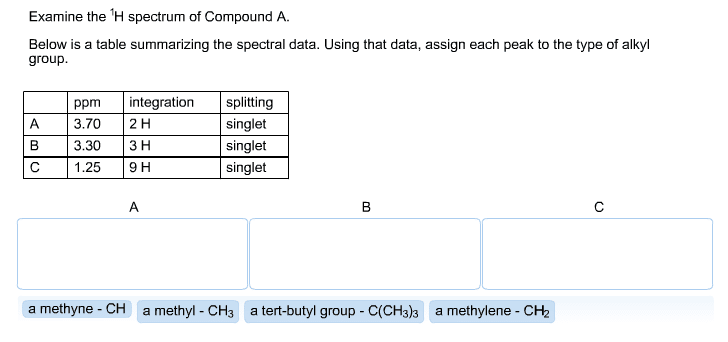
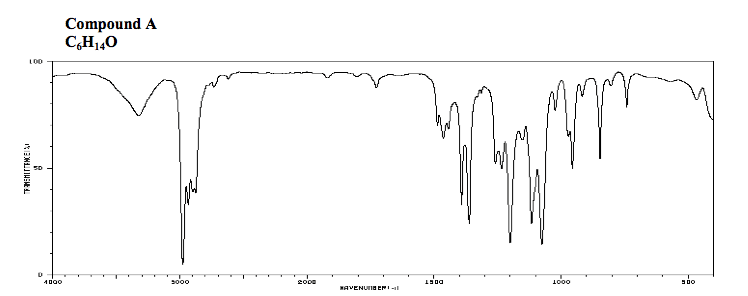
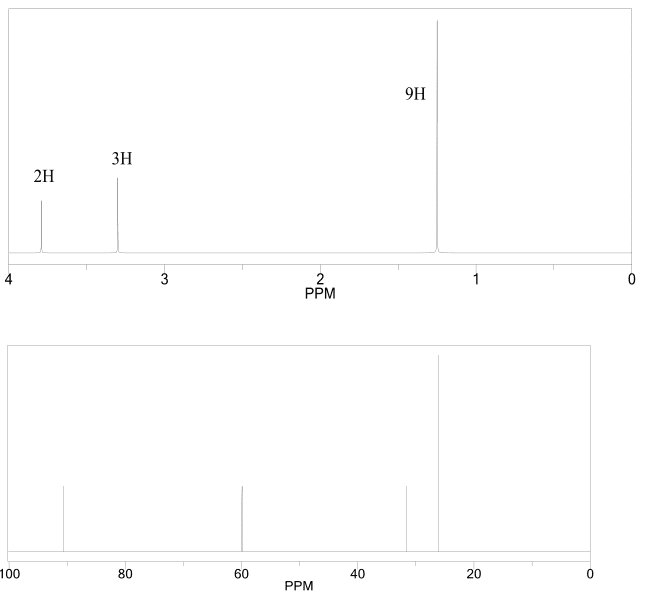
1
answer
0
watching
122
views
23 Nov 2019
Where can I find Good lecture notes for zumdahl 7th edition or zumdahl 8th edition chemistry book? Please make sure you check the link to the notes actually work, because I've searched hard and came up with nothing substantial.
Where can I find Good lecture notes for zumdahl 7th edition or zumdahl 8th edition chemistry book? Please make sure you check the link to the notes actually work, because I've searched hard and came up with nothing substantial.
1
answer
0
watching
122
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Keith LeannonLv2
30 May 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
Hi, so I am having a hard time creating the rate law because I can not figure out how to find the reaction order. I keep ending up with .5 = 5^x. Please help! The test is tomorrow
| Experiment | Initial Concentration CO (M) | Initial Concentration NO2 (M) | Initial Rate of Reaction (M/sec) |
|---|---|---|---|
| 1 | 5.0x10^-4 | 3.6x10^-5 | 3.4x10^-8 |
| 2 | 5.0x10^-4 | 1.8x10^-5 | 1.7x10^-8 |
| 3 | 1.0x10^-3 | 3.6x10^-5 | 6.8x10^-8 |
| 4 | 1.5x10^-3 | 7.2x10^-5 | ? |
A. What is the rate law for this reaction? Make sure to include the units for k in your work.
B. What is the initial rate of the reaction in Experiment 4?