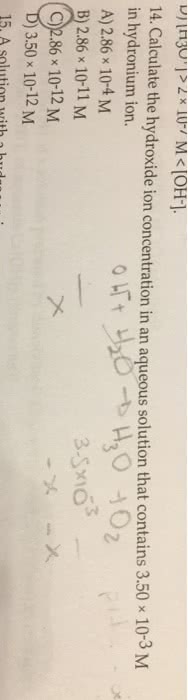1
answer
0
watching
97
views
27 Nov 2019
This might not be a good question to ask on here but I don't knowwhere else to try and get some help. What I cant figure out is whatbuttons to push on my calculater to get the right answer. I have aTI-36X Pro calculater. If anyone has any idea how I use it for thisproblem I would very much apreciate your help.
Determine the pH of an aqueous solution of CaCO3 that contains1.7*10^-6 M of hydroxide ion [OH^-].
(I need to get an answer of 8.2. This is the answer my teachergives. Others have helped me on here but gave me differentanswers.)
This might not be a good question to ask on here but I don't knowwhere else to try and get some help. What I cant figure out is whatbuttons to push on my calculater to get the right answer. I have aTI-36X Pro calculater. If anyone has any idea how I use it for thisproblem I would very much apreciate your help.
Determine the pH of an aqueous solution of CaCO3 that contains1.7*10^-6 M of hydroxide ion [OH^-].
(I need to get an answer of 8.2. This is the answer my teachergives. Others have helped me on here but gave me differentanswers.)
Beverley SmithLv2
26 Apr 2019