1
answer
0
watching
219
views
27 Nov 2019
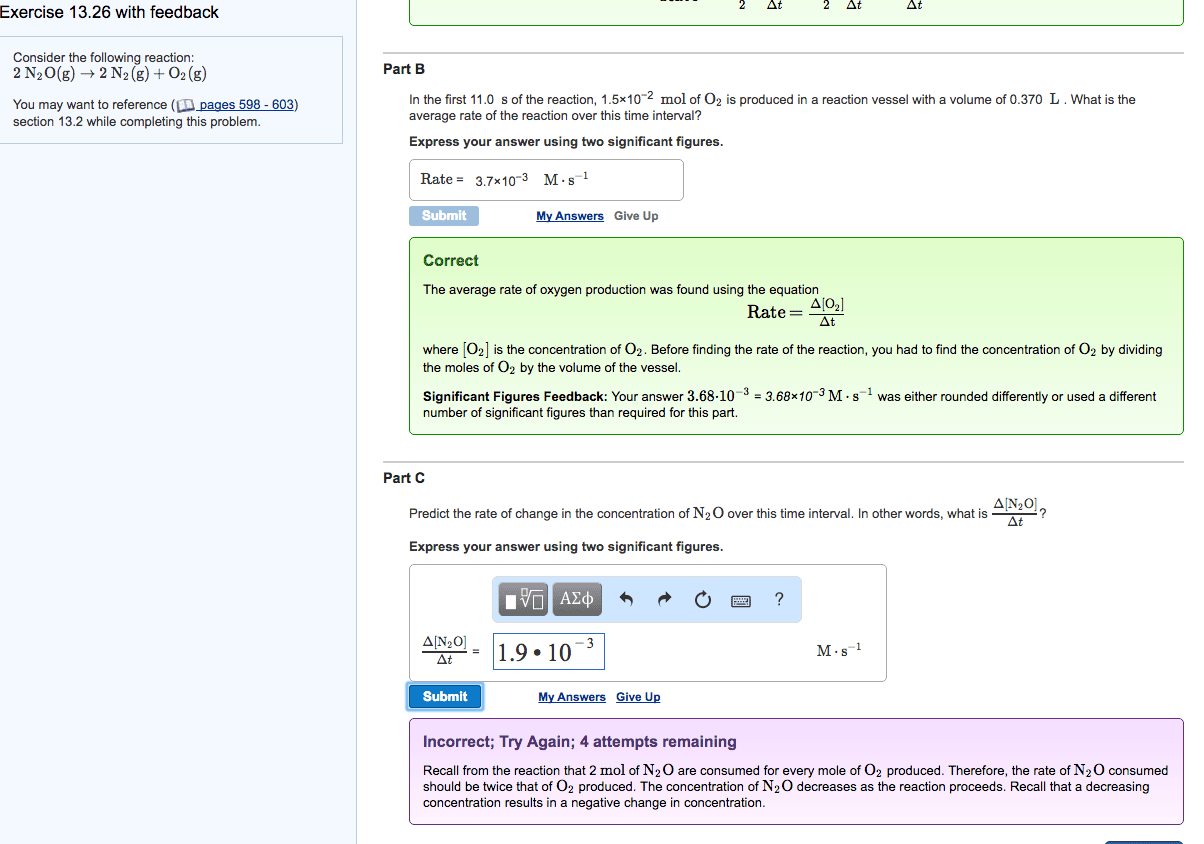
26. Consider the reaction:
2N2O (g) = 2N2 (g) + O2 (g)
A. Express the rate of the reaction with respect to each of thereactants and products.
B. In the first 15.0 s of the reaction, 0.015 mol of O2 is producedin a reaction vessel with a volume of 0.500 L. What is the averagerate of the reaction over this time interval.
C. Predict the rate of change in the concentration of N2O over thistime interval. In other words, what is [N2O]/t?
26. Consider the reaction:
2N2O (g) = 2N2 (g) + O2 (g)
A. Express the rate of the reaction with respect to each of thereactants and products.
B. In the first 15.0 s of the reaction, 0.015 mol of O2 is producedin a reaction vessel with a volume of 0.500 L. What is the averagerate of the reaction over this time interval.
C. Predict the rate of change in the concentration of N2O over thistime interval. In other words, what is [N2O]/t?
Lelia LubowitzLv2
13 Sep 2019