1
answer
0
watching
113
views
13 Dec 2019
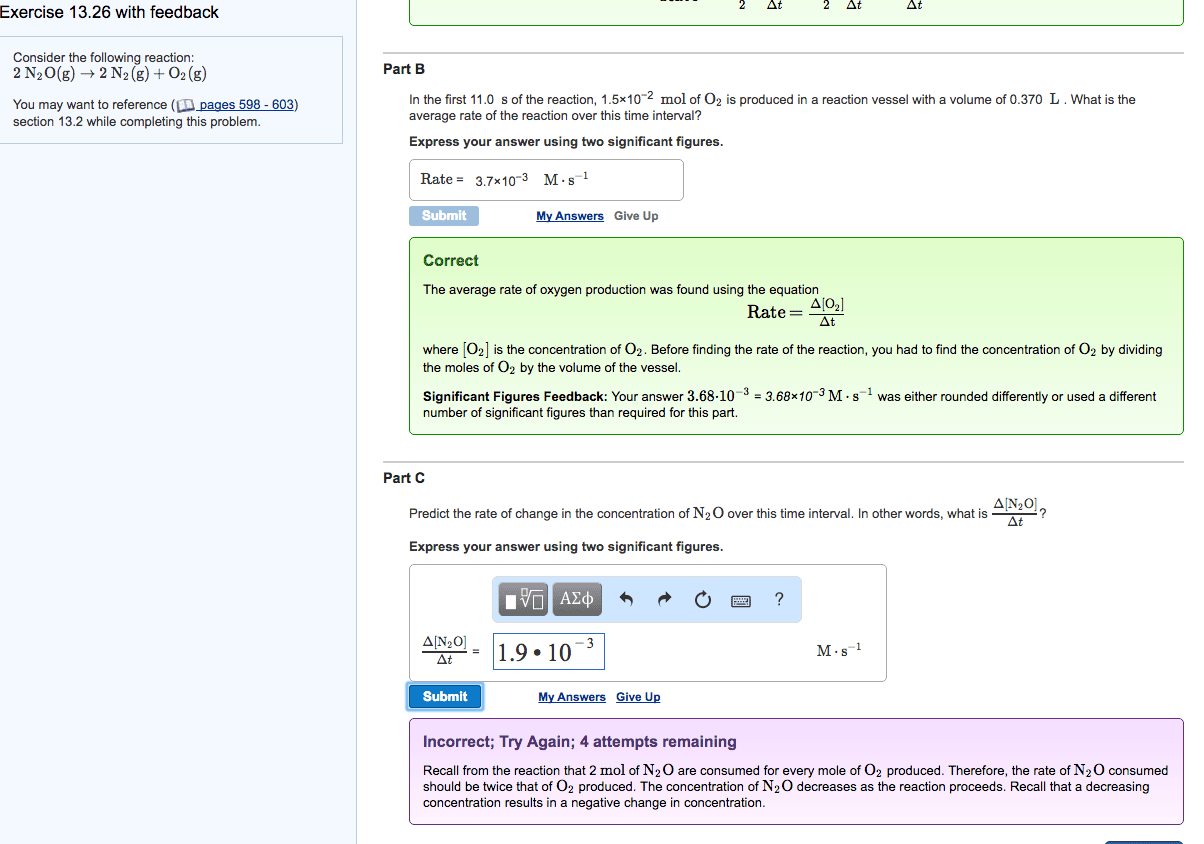
Consider the reaction 2N2O(g) ---> 2N2(g)+ O2(g)
a. In the first 10.0s of the reaction, 0.018 mol of O2 isproduce in a reaction vessel with a volume of 0.250L. What is theaverage rate of the reaction over this time interval?
b. Predict the rate of change in the concentration of N2O overthis time interval. In other words, what is dt [N2O]/dt?
d= delta
**Please show your work for I can understand how to do it.
Consider the reaction 2N2O(g) ---> 2N2(g)+ O2(g)
a. In the first 10.0s of the reaction, 0.018 mol of O2 isproduce in a reaction vessel with a volume of 0.250L. What is theaverage rate of the reaction over this time interval?
b. Predict the rate of change in the concentration of N2O overthis time interval. In other words, what is dt [N2O]/dt?
d= delta
**Please show your work for I can understand how to do it.
Jamar FerryLv2
17 Dec 2019