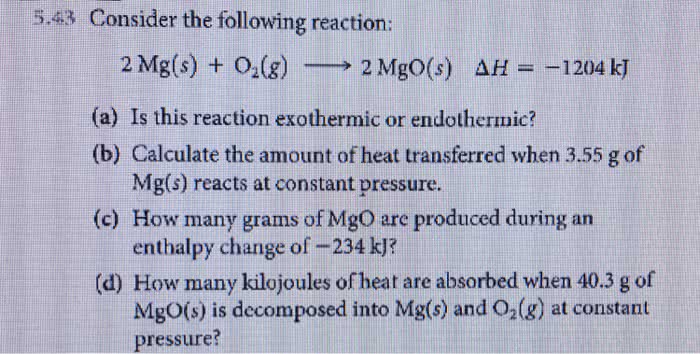1
answer
0
watching
674
views
11 Dec 2019
Consider the following reaction: 2Mg(s)+O2(g) → 2MgO(s), ΔH=−1204kJ. Is this reaction exothermic or endothermic? How do you calculate the amount of heat transferred when 2.4 grams of Mg(s) react at constant pressure?
Consider the following reaction: 2Mg(s)+O2(g) → 2MgO(s), ΔH=−1204kJ. Is this reaction exothermic or endothermic? How do you calculate the amount of heat transferred when 2.4 grams of Mg(s) react at constant pressure?
Alyssa Mae RemarimLv10
21 Oct 2020