1
answer
0
watching
375
views
11 Dec 2019
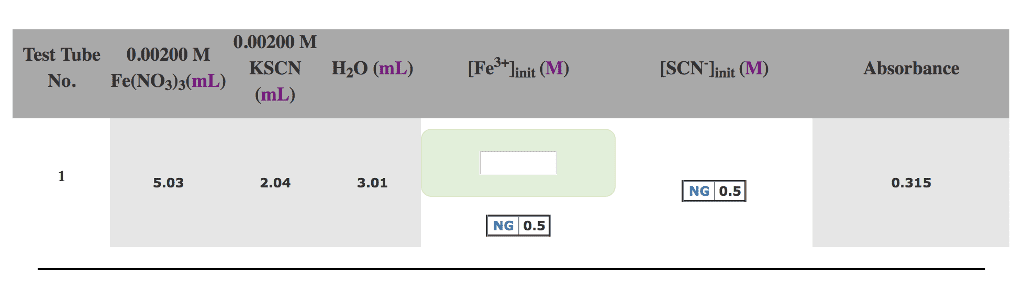
A solution is made up of
15.0 mL of 4.50 x 10^-3 M Fe(NO3)3
15.0 mL of 4.50 x 10^-3 M KSCN
10.0 mL of water
It is determined that the following reaction occurs
Fe3+ (aq) + SCN- (aq) = Fe(SCN)2+ (aq)
1. What is the concentration (in M) of Fe3+ in the resulting solution before any reaction takes place? Assume that volumes are addictive.
2. At equilibruim, [Fe(SCN)2+] is 1.20 x 10^-3 M. What is [Fe3+] at equilibrium?
3. What is K for the reaction at the temperature in which the experiment is performed?
A solution is made up of
15.0 mL of 4.50 x 10^-3 M Fe(NO3)3
15.0 mL of 4.50 x 10^-3 M KSCN
10.0 mL of water
It is determined that the following reaction occurs
Fe3+ (aq) + SCN- (aq) = Fe(SCN)2+ (aq)
1. What is the concentration (in M) of Fe3+ in the resulting solution before any reaction takes place? Assume that volumes are addictive.
2. At equilibruim, [Fe(SCN)2+] is 1.20 x 10^-3 M. What is [Fe3+] at equilibrium?
3. What is K for the reaction at the temperature in which the experiment is performed?
Jamar FerryLv2
13 Dec 2019