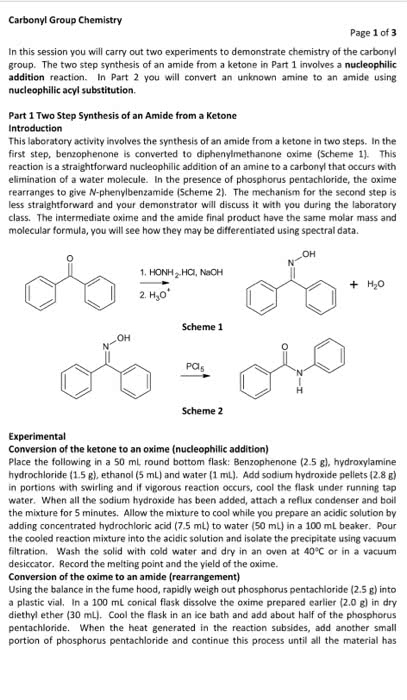
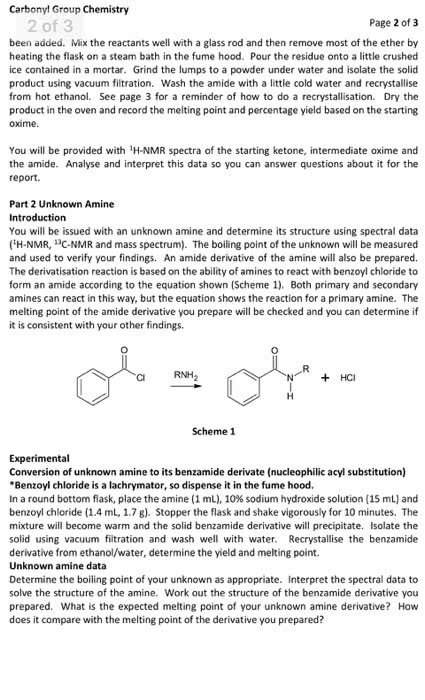
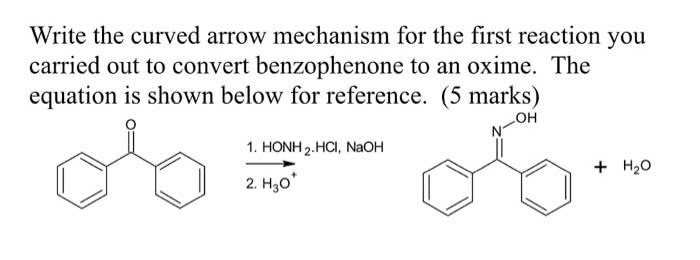
ACYLATION OF AN AROMATIC AMINE
Prepare a solution of 2.5 g of p-toluidine, 65 mL of water, and2.5 mL of concentrated
hydrochloric acid in a 125 mL Erlenmeyer flask. Be sure to placethe reagents in the flask in that
order! Using a warm hot plate to assist you, swirl the contentsof the flask until all of the solid
dissolves. The purpose of the acid is to protonate thep-toluidine and increases its solubility in
water.
In a small beaker, dissolve 3.75 g of sodium acetate trihydratein 7 mL of water. Again,
use a warm hot plate to speed up the process, if necessary. Thepurpose of the sodium acetate is
to buffer the solution. We want the pH such that the carbonylgroup is electrophilic but some of
the amine group is not protonated. A protonated carbonyl groupis more susceptible to
nucleophilic attack, while the amino group can only act as anucleophile when it is not
protonated.
Heat the p-toluidine hydrochloride solution to 50 ?C using a hotplate. While stirring or
swirling rapidly, add 2.5 mL of acetic anhydride from a burettein the hood. Then add the
sodium acetate solution prepared above. Once the solution isthoroughly mixed, cool the flask in
an ice-water bath. A white precipitate should form. Collect thissolid via vacuum filtration.
While the solid is on the filter paper, wash it three times withcold water. After washing the
solid, remove as much of the water from the product by suctionfiltration as possible. Once the
product is fully dry, determine the mass, measure the meltingpoint and calculate the percent
yield. Let your Teaching Assistant evaluate your product anddispose of it properly
please please please please help me do allthese:
calculate the limiting reagent and
the theoretical yield
and the chemical balanced equation
ACYLATION OF AN AROMATIC AMINE
Prepare a solution of 2.5 g of p-toluidine, 65 mL of water, and2.5 mL of concentrated
hydrochloric acid in a 125 mL Erlenmeyer flask. Be sure to placethe reagents in the flask in that
order! Using a warm hot plate to assist you, swirl the contentsof the flask until all of the solid
dissolves. The purpose of the acid is to protonate thep-toluidine and increases its solubility in
water.
In a small beaker, dissolve 3.75 g of sodium acetate trihydratein 7 mL of water. Again,
use a warm hot plate to speed up the process, if necessary. Thepurpose of the sodium acetate is
to buffer the solution. We want the pH such that the carbonylgroup is electrophilic but some of
the amine group is not protonated. A protonated carbonyl groupis more susceptible to
nucleophilic attack, while the amino group can only act as anucleophile when it is not
protonated.
Heat the p-toluidine hydrochloride solution to 50 ?C using a hotplate. While stirring or
swirling rapidly, add 2.5 mL of acetic anhydride from a burettein the hood. Then add the
sodium acetate solution prepared above. Once the solution isthoroughly mixed, cool the flask in
an ice-water bath. A white precipitate should form. Collect thissolid via vacuum filtration.
While the solid is on the filter paper, wash it three times withcold water. After washing the
solid, remove as much of the water from the product by suctionfiltration as possible. Once the
product is fully dry, determine the mass, measure the meltingpoint and calculate the percent
yield. Let your Teaching Assistant evaluate your product anddispose of it properly
please please please please help me do allthese:
calculate the limiting reagent and
the theoretical yield
and the chemical balanced equation
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
Procedure: Setting Up: Add 1.0 g of p-aminobenzoic acid and 10 mL of absolute ethanol to the round-bottom flask containing a stir bar. Stir the mixture until the solid is completely dissolved. Add 1 mL of concentrated [18M] sulfuric acid dropwise to the ethanolic solution of p-aminohenzoic acid, equip the flask with a condenser, and set up the apparatus for heating under reflux using the heating mantle. Reaction: Heat the mixture under gentle reflux for 30 min, stirring all the while. If any solid remains in the flask at this time, remove the flask from the heat source and allow the mixture to cool for 2-3 min. Add an extra 3 mL of ethanol and 0.5 mL of concentrated H2SO4, to the reaction flask and resume heating under reflux. After the reaction mixture becomes homogeneous, continue heating it under gentle reflux for another 30 min. For the experiment to succeed, it is important that all of the solids dissolve during the reflux period. Work-Up and Isolation: Allow the reaction mixture to cool to room temperature and then pour it into a beaker containing 30 mL of water. Basify the mixture to a pH of about 8 by slowly adding 10% aqueous sodium carbonate with stirring. Be careful in this step as frothing occurs during the neutralization. You should calculate beforehand the approximate volume of 10% aqueous sodium carbonate that will be required to neutralize the total amount of sulfuric acid you used. Collect the crude benzocaine that precipitates by vacuum filtration. Use three 10-mL portions of cold water to rinse the solid from the beaker and wash the filter cake. Air-dry the product. Purification: Weigh the crude product and recrystallize from methanol/water pair of solvents: Place the solid in a 50 mL Erlenmeyer flask. Add the minimum amount of hot methanol to dissolve it. Keep on a steam bath. Slowly add hot water to the solution using a disposable pipette just until the solution becomes cloudy and fails to clear up when it is swirled. To the hot solution on the steam bath add hot methanol, drop wise, until the turbidity or crystals just disappear. Allow the solution to cool to room temperature and then cool it in an ice-water bath for 10-15 min to complete crystallization. Isolate the crystals by vacuum filtration, wash them with 5-10 mL of cold water, and allow them to air-dry. Weigh the crystals and calculate the percentage yield.
True or False? When analyzing the IR spectra of starting material and product, a positive indication suggesting that the product was made would be the disappearance of a strong stretch between 1690-1740 cm-1.
|
Why is it important that all of the solids dissolve during the reflux period for you to obtain a good yield of product?
A. Because everything needs to be in solution for the reaction to happen to the fullest extent.
B. Because the product will be impure if the intermediate doesn't dissolve completely.
C. Because the ethanol will evaporate off if it's not dissolved.
D. All of the above
What is the gas that is evolved during neutralization?
A. O2
B. CO2
C. SO2
D. CO3
Why is it necessary to neutralize the reaction mixture to work up the product?
A. Because it is dangerous to work with an acidic solution.
B. Because after the addition of ethanol and sulfuric acid, the benzocaine exists as ammonia and must be protonated so that it becomes insoluble and collectable.
C. Because after the addition of ethanol and sulfuric acid, the benzocaine exists as a soluble carboxylic acid and must be neutralized so that it becomes insoluble and collectable.
D. Because after the addition of ethanol and sulfuric acid, the benzocaine exists as a soluble ammonium salt and must be neutralized so that it becomes insoluble and collectable.
If you end up adding 1.5 mL of concentrated sulfuric acid (18 M), how many mL of 10% aqueous sodium carbonate would be required to neutralize the reaction mixture?
A. 1.5 mL
B. 3 mL
C. 15 mL
D. 29 mL
What is the purpose of the sulfuric acid?
A. To catalyze the reaction.
B. To protonate the intermediate salt.
C. To protonate the ethanol.
D. To increase the pH of the reaction mixture.
What is the theoretical yield of the product?
A. 1.00 g
B. 2.98 g
C. 1.20 g
D. 10 mL