0
answers
1
watching
397
views
13 Dec 2019
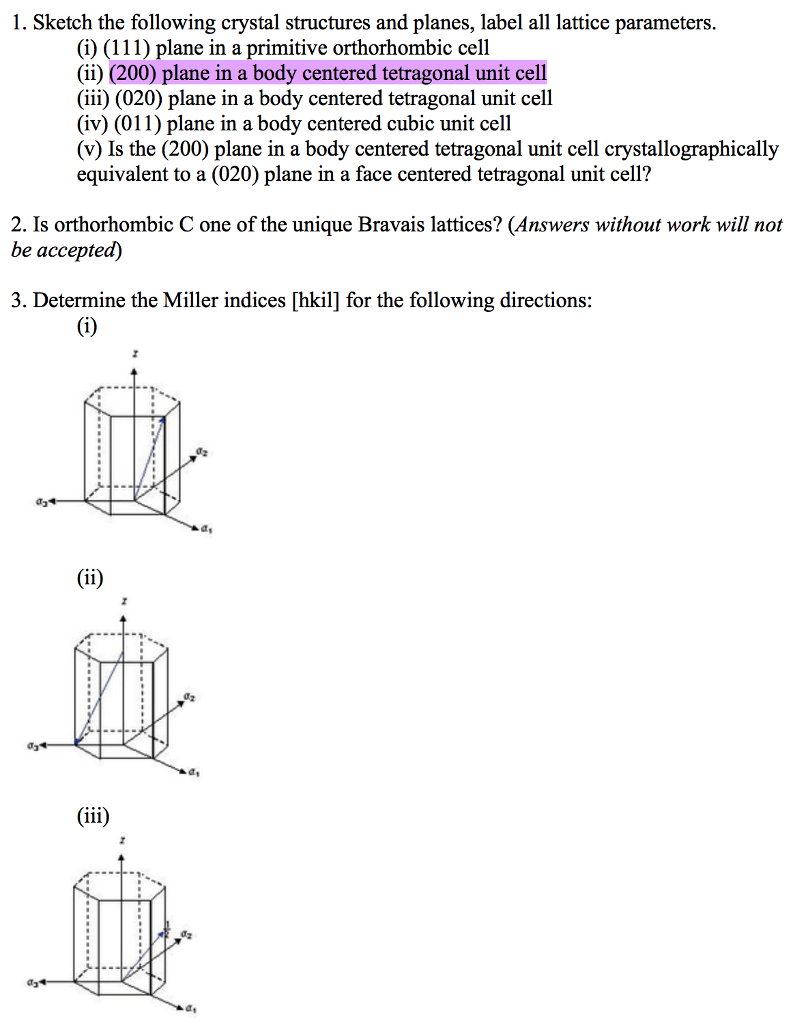
1) Calculate the lattice parameter of KI
2) Determine the number of K and I ions in the KI unit cell
3) Determine KI's bulk density
4) Sketch the (020) plane of KI
5) Calculate the planar density of the (020) plane of KI, expressed as a decimal
1) Calculate the lattice parameter of KI
2) Determine the number of K and I ions in the KI unit cell
3) Determine KI's bulk density
4) Sketch the (020) plane of KI
5) Calculate the planar density of the (020) plane of KI, expressed as a decimal
0
answers
1
watching
397
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
1.) Assume that each drop of Pb(NO3)2 contains 1.7 mg of Pb(NO3)2 and that each drop of KI contains 0.83 mg of KI. Given this information, determine the limiting reagent and calculate the theoretical yield for each reaction Tube 1-5 listed in the table below.
Table 1.1
| Drops of water | Drops of Pb(NO3)2 | Drops of KI | Lead:Iodine Ration | ||
| Tube 1 | 32 | 12 | 4 | 3:1 | |
| Tube 2 | 30 | 12 | 6 | 2:1 | |
| Tube 3 | 24 | 12 | 12 | 1:1 | |
| Tube 4 | 12 | 12 | 24 | 1:2 | |
| Tube 5 | 0 | 12 | 36 | 1:3 |