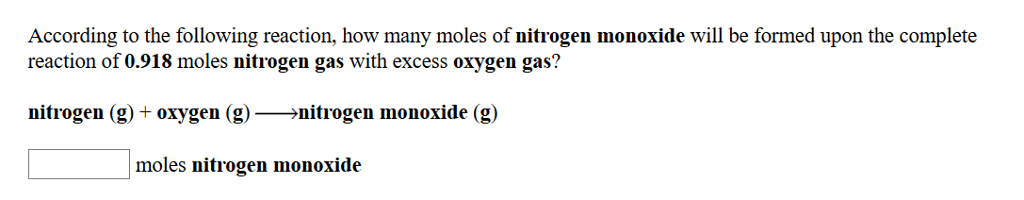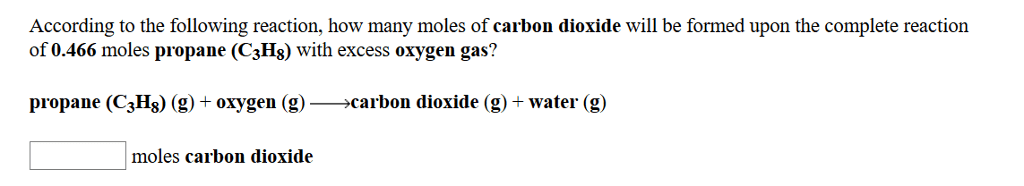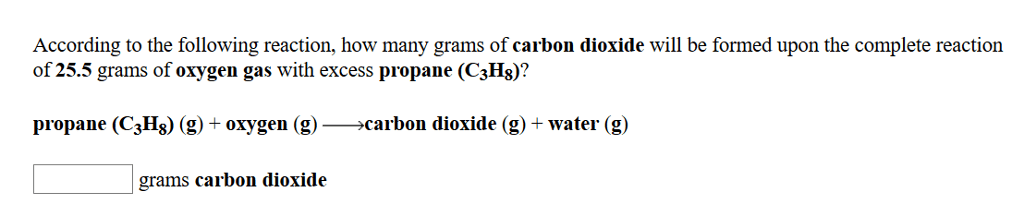1
answer
0
watching
67
views
13 Dec 2019
In the reaction 2 N2+ O2-> 2 N2O,if starting with 0.644 moles of nitrogen and 0.319 moles of oxygen,how many moles of dinitrogen monoxide can be made? (Hint: this maybe a limiting reactant case)
In the reaction 2 N2+ O2-> 2 N2O,if starting with 0.644 moles of nitrogen and 0.319 moles of oxygen,how many moles of dinitrogen monoxide can be made? (Hint: this maybe a limiting reactant case)
Nelly StrackeLv2
17 Dec 2019