1
answer
0
watching
427
views
13 Dec 2019
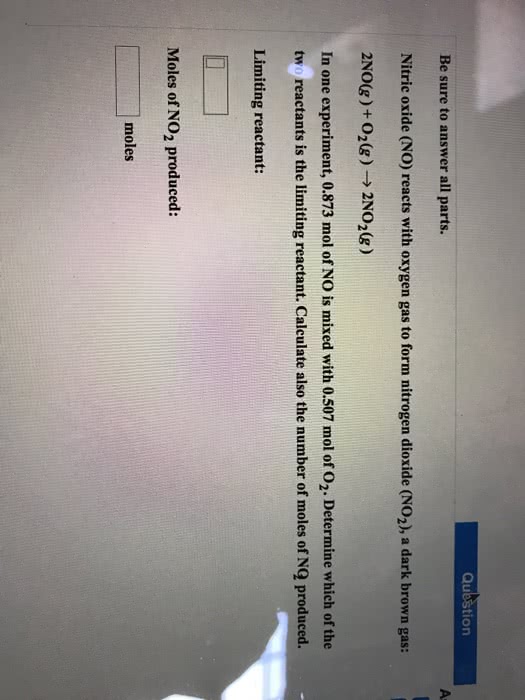
nitric oxide reacts with oxygen gas to form nitrogen dioxide adark brown gas: 2NO(g)+O2(g)--->2NO2 (g)
in one experiment 0.861 mol of NO is mixed with 0.500 mol of O2.determine which of the two reactants is the limiting reactant andcalculate the number of NO2 moles produced
nitric oxide reacts with oxygen gas to form nitrogen dioxide adark brown gas: 2NO(g)+O2(g)--->2NO2 (g)
in one experiment 0.861 mol of NO is mixed with 0.500 mol of O2.determine which of the two reactants is the limiting reactant andcalculate the number of NO2 moles produced
Lelia LubowitzLv2
17 Dec 2019