1
answer
0
watching
134
views
18 Dec 2019
1.) How many grams of Mg(OH)2 are required to obtain a 0.01M hydroxide solution?
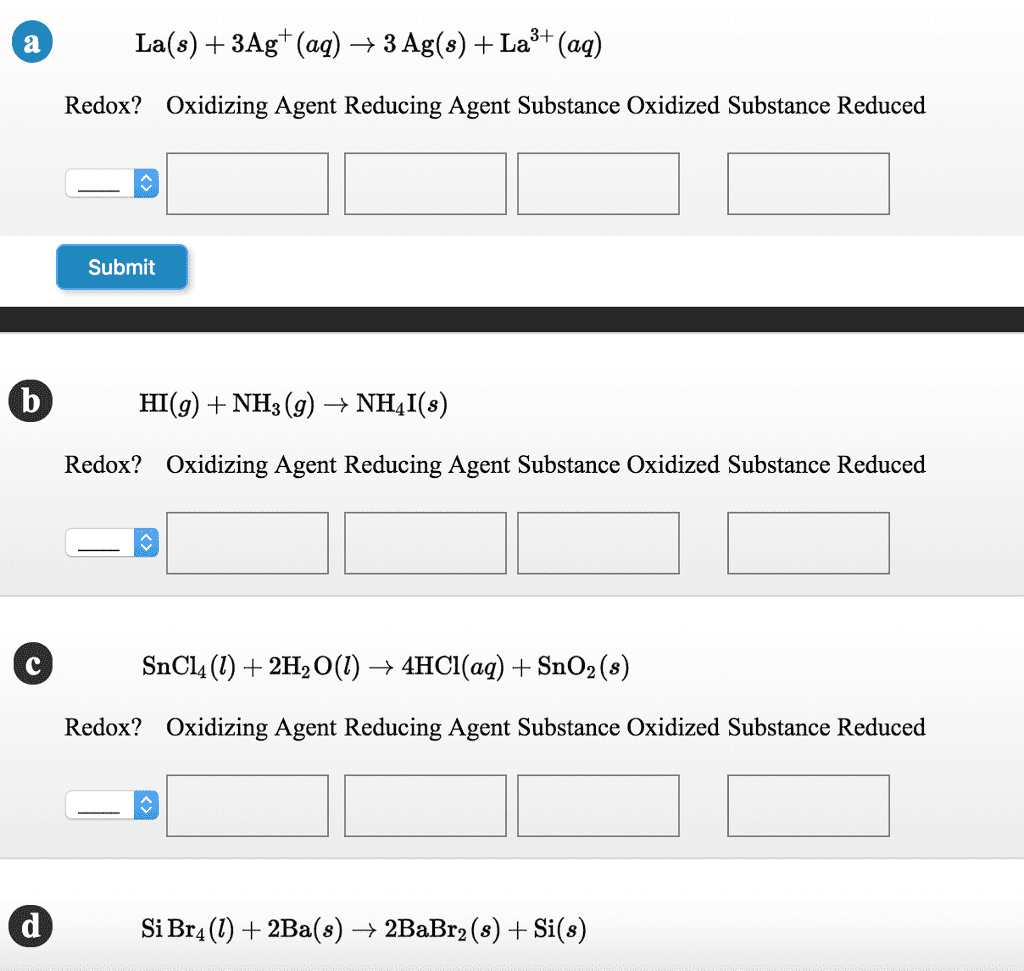
2) Specify which of the following are redox reactions and identify the oxidizing agent, the reducing agent, the substance being oxidized, and the substance being reduced.
a) Ba(s) + MgCl2(aq) --> Mg(s) + BaCl2(aq)
b) HNO3(aq) + NH4OH(aq) --> NH4NO3(aq) + H2O(l)
c) SiCl4(l) + 2Ca(s) --> 2CaCl2(s) + Si(s)
d) 2Cr(s) + 6HCl(aq) --> 2CrCl3(s) + 3H2(g)
1.) How many grams of Mg(OH)2 are required to obtain a 0.01M hydroxide solution?
2) Specify which of the following are redox reactions and identify the oxidizing agent, the reducing agent, the substance being oxidized, and the substance being reduced.
a) Ba(s) + MgCl2(aq) --> Mg(s) + BaCl2(aq)
b) HNO3(aq) + NH4OH(aq) --> NH4NO3(aq) + H2O(l)
c) SiCl4(l) + 2Ca(s) --> 2CaCl2(s) + Si(s)
d) 2Cr(s) + 6HCl(aq) --> 2CrCl3(s) + 3H2(g)
Nelly StrackeLv2
31 Dec 2019