1
answer
0
watching
530
views
5 Apr 2020
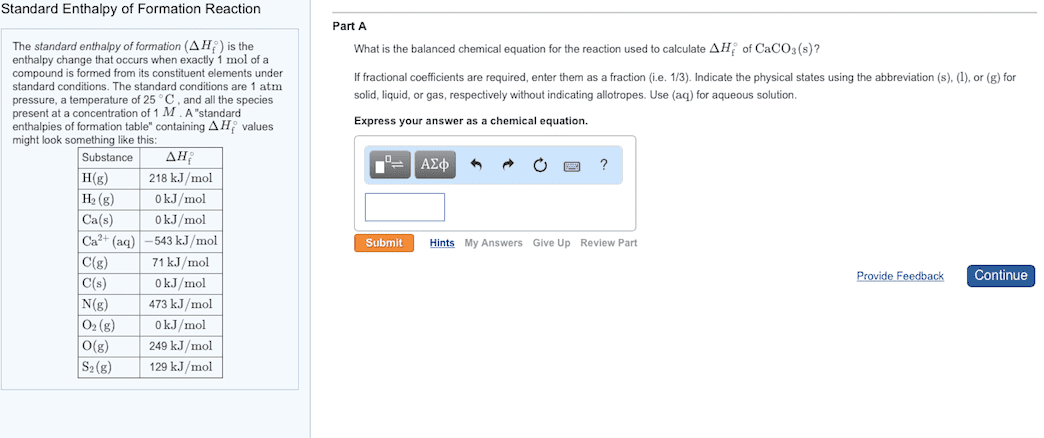
(a) Why are tables of standard enthalpies of formation so useful? (b) What is the value of the standard enthalpy of formation of an element in its most stable form? (c) Write the chemical equation for the reaction whose enthalpy change is the standard enthalpy of formation of sucrose (table sugar), C12H22O11(s), ∆Hf°[C12H22O11].
(a) Why are tables of standard enthalpies of formation so useful? (b) What is the value of the standard enthalpy of formation of an element in its most stable form? (c) Write the chemical equation for the reaction whose enthalpy change is the standard enthalpy of formation of sucrose (table sugar), C12H22O11(s), ∆Hf°[C12H22O11].
Bunny GreenfelderLv2
20 May 2020